Your shares will be counted towards the quorum only if you submit a valid proxy (or one is submitted on your behalf by your broker, bank or other nominee) or if you vote in persononline at the meeting. Abstentions and brokernon-votes will be counted towards the quorum requirement. If there is no quorum, either the Chair of the Board, without a stockholder vote, or the stockholders so present, by a majority in voting power thereof, may adjourn the meeting to another date without a stockholder vote.
date.
How can I find out the results of the voting at the Annual Meeting?
Preliminary voting results will be announced at the Annual Meeting. In addition, final voting results will be published in a current report on Form
8-K that we expect to file within four business days after the Annual Meeting. If final voting results are not available to us in time to file a
Form 8-K within four business days after the meeting, we intend to file a
Form 8-K to publish preliminary results and, within four business days after the final results are known to us, file an additional
Form 8-K to publish the final results.
When are stockholder proposals and director nominations due for next year’s Annual Meeting?
To be considered for inclusion in next year’s proxy materials, your proposal must be submitted in writing by December
27, 2019,13, 2021, to our
Corporate Secretary at
501 Canal7000 Marina Boulevard,
Richmond,Brisbane, California
94804,94005, and you must comply with all applicable requirements of Rule
14a-8 promulgated under the Securities Exchange Act of 1934, as amended, or the Exchange Act.
However, if our 2022 Annual Meeting of Stockholders is not held between April 25, 2022 and June 24, 2022, then the deadline will be a reasonable time prior to the time that we begin to print and mail our proxy materials.
Pursuant to our Bylaws, if you wish to bring a proposal before the stockholders or nominate a director at the
20202022 Annual Meeting of
stockholders,Stockholders, but you are not requesting that your proposal or nomination be included in next year’s proxy materials, you must notify our
Corporate Secretary, in writing, not later than the close of business on
March 12, 2020February 24, 2022 nor earlier than the close of business on
February 11, 2020.January 25, 2022. However, if our
20202022 Annual Meeting of
stockholdersStockholders is not held between
May 11, 2020April 25, 2022 and
July 10, 2020,June 24, 2022, to be timely, notice by the stockholder must be received not later than the 10th day following the day on which the first public announcement of the date of the
20202022 Annual Meeting is made or the notice of the meeting is mailed, whichever first occurs. You are also advised to review our Bylaws, which contain additional requirements about advance notice of stockholder proposals and director nominations.
The chair of the Annual Meeting may determine, if the facts warrant, that a matter has not been properly brought before the meeting and, therefore, may not be considered at the meeting. In addition, the proxy solicited by the Board for the
20202022 Annual Meeting of Stockholders will confer discretionary voting authority with respect to any proposal (i) presented by a stockholder at that meeting for which we have not been provided with timely notice and (ii) made in accordance with our Bylaws, if (x) the
20202022 proxy statement briefly describes the matter and how management’s proxy holders intend to vote on it, and (y) the stockholder does not comply with the requirements of Rule
14a-4(c)(2) promulgated under the Exchange Act.
Our Board currently consists of nine
directors. However, each of Steven J. Mento, Ph.D.directors and
Roger Jeffs, Ph.D. have determined not to stand forre-election at the Annual Meeting and their terms will expire as of the Annual Meeting. Accordingly, there are
sevennine nominees for director this year. Proxies may not be voted for a greater number of persons than the number of nominees named in this Proxy Statement. Each director to be elected and qualified will serve until the next annual meeting of stockholders
orand until a successor for such director is duly elected and qualified, or until the
earlier death, resignation or removal of such director. The nominees for election have agreed to serve if elected, and management has no reason to believe that such nominees will be unavailable to serve. In the event the nominees are unable or decline to serve as directors at the time of the Annual Meeting, the proxies will be voted for any nominee who may be designated by the present Board of Directors to fill the vacancy. Unless otherwise instructed, the proxy holders will vote the proxies received by them FOR the nominees named below.
Each of the
sevennine nominees, other than
Karen L. Smith, M.D., Ph.D.,Dr. Kenneth J. Hillan, has been previously elected by
theour stockholders. Dr.
Smith’s candidacyHillan was
initially recommended to the Nominating and Corporate Governance Committee
of the Board, or the Nominating and Governance Committee, by our Chief Executive Officer
upon the recommendation of another Board Director. Following this recommendation, the Board interviewed and
another current director.vetted Dr. Hillan prior to electing him to the Board. While the Nominating and Corporate Governance Committee and the Board chose to nominate
Dr. Alexander D. Macrae, our Chief Executive Officer, for election to the Board for the reasons set forth in his biography below, Dr. Macrae’s employment agreement also provides that the Board shall nominate Dr. Macrae for election as a member of the Board at each annual meeting of stockholders occurring while Dr. Macrae’s employment agreement is in effect.
Each of the
sevennine nominees set forth in this Proxy Statement will be elected by the majority of the votes cast with respect to such nominee, which
shall meanmeans that the number of
votes “For”
votes a nominee’s election
exceedsmust exceed the number of
votes “Against”
votesthat nominee (with abstentions and broker
non-votes not counted as either a “For” or “Against” vote for that nominee’s election). If an incumbent director does not receive the required majority vote, the director
shallis required under our Bylaws to promptly tender his or her resignation to the Board. Within 90 days after the Annual Meeting, the Nominating and Corporate Governance Committee
of the Board will make a recommendation to the Board
of Directors as to whether to accept or reject the resignation. The Board will act by taking into account such committee’s recommendation. If the Board does not accept the resignation, the Board is required to publicly disclose its decision and the rationale behind the decision.
The following includes a brief biography of each nominee for director, including their respective ages as of
April 25, 2019.March 31, 2021. Each biography includes information regarding the specific experience, qualifications, attributes or skills that led the Nominating and Corporate Governance Committee and the Board to determine that the applicable nominee should serve as a member of the
Board as of the date of this proxy statement.Board.
Alexander D. Macrae, M.B., Ch.B., Ph.D., age 56,58, has served as our President and Chief Executive Officer and as a member of our Board of Directors since June 2016. Dr. Macrae also serves on the board of directors of 4D Pharma plc, a biopharmaceutical company. He has over twenty20 years of experience in the pharmaceutical industry most recently serving as the Global Medical Officer of Takeda Pharmaceuticals,Pharmaceutical Company Limited, or Takeda, a public global biopharmaceutical company, from 2012 to March 2016, where he established and led the Global Medical Office, which encompassesencompassed medical affairs, regulatory affairs, pharmacovigilance, outcomes research and epidemiology, quantitative sciences and knowledge and informatics. From 2001 to 2012, Dr. Macrae held roles of increasing responsibility at GlaxoSmithKline plc, or GSK, a public global healthcare company, including Senior Vice President, Emerging Markets Research and Development, or R&D, from 2009 to 2012. In that position, he provided expertise and resources to create afirst-of-its-kind group to expand GSK’s global reach by providing R&D strategies, clinical development and regulatory resources to enter emerging markets and Asia-Pacific. From 2007 to 2008, he was Vice President, Business Development. In that7
position, he was responsible for scientific assessment and business development project leadership for the neurology, psychiatry, cardiovascular and metabolic therapeutic areas. Earlier in his career, he worked for SmithKline Beecham, where he was responsible for clinical development in the therapeutic areas of neurology and gastroenterology. Dr. Macrae received his B.Sc. in pharmacology and his M.B., Ch.B. with honors from Glasgow University. He is a member of the Royal College of Physicians. Dr. Macrae also earned his Ph.D. in molecular genomics at King’s College, Cambridge. The Nominating and Corporate Governance Committee and the Board believe that Dr. Macrae’sday-to-day leadership and intimate knowledge of our business and operations, as well as our relationships with partners, collaborators and investors, provides the Board with anin-depth understanding of the Company.
Robert F. Carey, age 60,62, has served as a member of our Board of Directors since June 2016. Mr. Carey has been executive viceserved as co-founder, president and chief businessoperating officer of ACELYRIN, Inc., a privately-held biopharmaceutical company, since July
2020. Mr. Carey served as Executive Vice President, Chief Business Officer for Horizon
PharmaTherapeutics plc, a biopharmaceutical company,
sincefrom March
2014.2014 to October 2019. Prior to that, he served as managing director and head of the healthcare investment banking group at JMP Securities LLC, a full-service investment bank from
March 2003 to
March 2014. Prior to JMP, Mr. Carey was a managing director in the healthcare groups at Dresdner Kleinwort Wasserstein and Vector Securities International, Inc. He has also held roles at Shearson Lehman Hutton and Ernst & Whinney. Mr. Carey
hasalso serves on the board of directors of Beyond Air, Inc. (formerly AIT Therapeutics, Inc.), a public medical device and biopharmaceutical company. Mr. Carey previously served on the board of directors of
ArgosFS Development Corp., a public blank-check company now known as Gemini Therapeutics, Inc.,
a life science company, since September 2015.from July 2020 to February 2021. Mr. Carey received his
B.S.BBA in
accountingaccountancy from the University of Notre Dame. The Nominating and Corporate Governance Committee and the Board believe that Mr. Carey’s extensive experience and knowledge in the healthcare investment banking industry, particularly with financings, global expansion and other strategic transactions by life-science companies, as well as his role in
senior management and business development for a public biopharmaceutical company, provides the Board with valuable insight and contribution.
Stephen G. Dilly, M.B.B.S.
Kenneth J. Hillan, M.B., Ph.D.Ch.B., age 59,60, has served as a member of our Board of Directors since March 2010.September 2020. Since May 2014,February 2019, Dr. DillyHillan has served as Head of Therapeutics of 23andMe, Inc., a privately-held consumer genetics and research company. From October 2011 to December 2017, Dr. Hillan was Chief Executive Officer of Achaogen, Inc., a public biopharmaceutical company, where he was President and President of R&D from January 2018 to October 2018 and a member of the boardBoard of directorsDirectors from October 2011 to October 2018. In April 2019, Achaogen filed a voluntary petition for relief under Chapter 11 of Aimmune Therapeutics,the U.S. Bankruptcy Code and most of its assets were subsequently sold to a multinational pharmaceutical company. Previously, Dr. Hillan worked at Genentech, Inc. (formerly Allergenfrom 1994 to 2011, where he led the medical and scientific strategies for its Immunology, Tissue Growth and Repair, or ITGR, drug portfolio, and held a number of key leadership positions in research and development, including Senior Vice President of Clinical Development, Inflammation; Vice President of ITGR; Vice President of Development Sciences; and Vice President of Research Corporation), a biotechnology company developing treatments for food allergies. From January 2012 to December 2012, he was Chief Executive Officer of PhotoThera, Inc., a medical device company. From 2006 to December 2011, heOperations and Pathology. He also served as Genentech’s Senior Vice President and Chief Executive OfficerHead of Clinical Development and a member ofProduct Development Strategy in Asia-Pacific for Roche in Shanghai, China. He also serves on the board of directors of APT Pharmaceuticals,Zymeworks, Inc., a drug developmentpublic biotechnology company. From 2007 to 2009, heDr. Hillan previously served as a member ofon the board of directors of Avigen,Relypsa, Inc., a biopharmaceutical company that merged with MediciNova, Inc. in December 2009. From 2003 from June 2014 to 2006, he served as Chief Medical Officer and Senior Vice President of Development of Chiron BioPharma, a biotechnology company thatSeptember 2016, when it was later acquired by Novartis InternationalGalenica AG. From 1998 to 2003, he held various management positionsDr. Hillan received his M.B. Ch.B. (Bachelor of Medicine and Surgery) degree from the Faculty of Medicine at Genentech, Inc., including Vice President of Development Sciences from 2002 to 2003 and Vice President of Medical Affairs from 1998 to 2001. From 1988 to 1998, Dr. Dilly held various management positions in drug development with SmithKline Beecham in the U.K. During his career, Dr. Dilly has been closely associated with the development and launch of marketed drugs for many therapeutic areas, including Kytril, Paxil, Kredex, Requip, TNKase, Xolair, Avastin, Raptiva, Tarceva, Lucentis and Cubicin. In 1982 Dr. Dilly received an M.B.B.S., the equivalent of an M.D. in the United States, from the University of London in the U.K. and a Ph.D. in cardiac physiology from University of London in 1988. Dr. Dilly brings medical expertise and significant drug development experience to our Board.Glasgow, United Kingdom. The Nominating and Corporate Governance Committee and the Board believe that Dr. Dilly’sHillan’s extensive experience and knowledge in all stagesthe development of drug development, from project prioritization through clinical trial designtherapeutics and data analysis to product launch,in the biotechnology industry provides the Board with valuable insight into this processand contribution to the Company’s development of genomic medicines.
John H. Markels, Ph.D., age 55, has served as a member of our Board of Directors since February 2020. Dr. Markels has over 30 years of leadership experience in the Company continuespharmaceutical industry. Since January 2019, he has served as President of Global Vaccines at Merck & Co., Inc., where he leads an integrated team dedicated to advancediscovery and developdevelopment, supply and access, and global marketing and long-term strategy for the vaccines portfolio. Earlier roles at Merck included President, Latin America from January 2018 to January 2019, SVP, Global Human Health Business Strategy from January 2017 to December 2017, Managing Director, Mexico from November 2013 to January 2017. Prior to his commercial roles, he had a long career in senior leadership positions in global manufacturing, including operations leadership for Europe, Middle East and Africa, Asia Pacific and emerging markets as well as manufacturing technology and product development roles in pharmaceutical active pharmaceutical ingredients (API) and vaccines. Throughout his tenure at Merck, Dr. Markels has led multiple enterprise level strategy efforts including the Merck manufacturing strategy, portfolio strategy, small molecule commercialization strategy and others. Dr. Markels received his Ph.D. in chemical engineering from the University of California, Berkeley and his B.S. in chemical engineering from the University of Delaware. The Nominating and Corporate Governance Committee and the Board believe that Dr. Markels’ extensive leadership experience in operations, strategy and development provides valuable operational, strategy and management skills to the Board.
James R. Meyers, age 56,has served as a member of our pipelineBoard of novel genomicDirectors since November 2019. He has over 30 years of commercial leadership experience in the biotechnology industry. Since November 2020, Mr. Meyers has served as President and cell therapy assets.Chief Executive Officer of IntraBio Ltd., a privately-held biopharmaceutical company. Previously, Mr. Meyers held positions of increasing responsibility at Gilead Sciences, Inc., or Gilead, from 1996 to February 2018, most recently as Gilead’s Executive Vice President of Worldwide Commercial Operations from November 2016 to February 2018, where he was responsible for global commercial activities, including pricing and market access in North America, Europe, Middle East, Australia and Japan. Mr. Meyers joined Gilead in 1996 and successfully led 25 product launches in seven different therapeutic areas over a 22-year period, including 11 brands with peak annual revenue greater than $1 billion. Prior to Gilead, Mr. Meyers held positions of increasing responsibility with Zeneca Pharmaceuticals and Astra USA. He currently serves on the board of directors of two other public biopharmaceutical companies, Arbutus Biopharma Corporation and CytomX Therapeutics, Inc., and he remains an active advisor to several major biopharmaceutical companies. Mr. Meyers achieved his B.S. in Economics from Boston College. The Nominating and Corporate Governance Committee and the Board believe that Mr. Meyers’ extensive
commercial leadership experience in the biotechnology industry provides valuable operational, commercial assessment and management skills to the Board.
H. Stewart Parker, age 63,65, has been a member of our Board of Directors since June 2014, and has been Chair since June 2017. Ms. Parker has over 3040 years of experience in the biotechnology industry. Prior to focusing on boards and consulting, she served as the Chief Executive Officer of The Infectious Disease Research Institute (IDRI), anot-for-profit global health Research institute from March 2011 to December 2013. In 1992, Ms. Parker founded Targeted Genetics Corporation, a publicly traded Seattle-based biopharmaceutical company formed to develop gene-based treatments for acquired and inherited diseases that became a world leader in adeno-associated virus, or AAV,8
gene therapy. She held the position of President and Chief Executive Officer and was a member of its board of directors from the company’s inception until November 2008. Prior to founding Targeted Genetics, Ms. Parker served in various capacities at Immunex from August 1981 through December 1991, most recently as Vice President, Corporate Development. From February 1991 to January 1993, Ms. Parker served as President and a Director of Receptech Corporation, a company formed by Immunex in 1989 to accelerate the development of soluble cytokine receptor products. She has served on the board of directors and the executive committee of BIO, the primary trade organization for the biotechnology industry. She currently serves as a member ofon the board of directors for severalfor-profit companies including C3J Therapeutics (formerly C3 Jian Inc),of Achieve Life Sciences, Inc., StrideBio Inc.a public pharmaceutical company and Nexgenia andpreviously served on the advisory boardsboard of the Universitydirectors of Washington Foundation and College of Arts & Sciences.Armata Pharmaceuticals, Inc., a public biotechnology company, from May 2019 to December 2020. Ms. Parker received her B.A. and M.B.A. from the University of Washington. The Nominating and Corporate Governance Committee and the Board believe that Ms. Parker’s senior executive experience in AAV gene therapy and biotechnology drug development provides valuable operational, commercial assessment and management skills to the Board.
Saira Ramasastry, age 43,45, has served as a member of our Board of Directors since June 2012. Since April 2009, she has served as Managing Partner of Life Sciences Advisory, LLC, a company that she founded to provide strategic advice, business development solutions and innovative financing strategies for the life science industry. Ms. Ramasastry also serves on the Industry Advisory Board of the Michael J. Fox Foundation for Parkinson’s Research and as business and sustainability lead for the European Prevention of Alzheimer’s Dementia consortium. From August 1999 to March 2009, Ms. Ramasastry was an investment banker with Merrill Lynch & Co., Inc. where she helped establish the biotechnology practice and was responsible for origination of mergers and acquisitions, or M&A, strategic and capital markets transactions. Prior to joining Merrill Lynch, she served as a financial analyst in the M&A group at Wasserstein Perella & Co., an investment banking firm, from July 1997 to September 1998. Ms. Ramasastry currently serves on the Industry Advisory Boardboard of the Michael J. Fox Foundation for Parkinson’s Research,directors of Vir Biotechnology, Inc., a public biotechnology company, Glenmark Pharmaceuticals, Ltd., a public pharmaceutical company, and Akouos, Inc., a public genetic medicine company. Ms. Ramasastry previously served on the board of directors of Pain Therapeutics,Innovate Biopharmaceuticals, Inc. (now 9 Meters Biopharma, Inc.), a public biopharmaceutical company, from June 2018 to March 2020, and Cassava Sciences, Inc., and lead business advisor for the European Prevention of Alzheimer’s Dementia consortium.a public biopharmaceutical company, from February 2013 to June 2020. Ms. Ramasastry received her B.A. in economics with honors and distinction and an M.S. in management science and engineering from Stanford University, as well as an M. Phil. in management studies from the University of Cambridge where she is a guest lecturer for the Bioscience Enterprise Programme and serves on the Cambridge Judge Business School Advisory Council. Ms. Ramasastry is also a Health Innovator Fellow of the Aspen Institute and a member of the Aspen Global Leadership Network. The Nominating and Corporate Governance Committee and the Board believe that Ms. Ramasastry’s extensive experience in global healthcare investment banking and strategic advisory consulting provides valuable financial, commercial assessment and business development skills to the Board and her thorough understanding of our technology and programs provides the Board with valuable insight in the development of our novel genomic and cell therapy assets.
Karen L. Smith, M.D., Ph.D., M.B.A., L.L.M., age 51,53, has served on our Board of Directors since June 2018. Dr. Smith is a life sciences thought leader with over 20 years of biopharmaceutical experience bringing drugs into the clinic and through commercialization. She has been a key contributor to the successful development of multiple FDA and EMA approved products in several therapeutic areas, including oncology (Herceptin, Vyxeos), rare disease (Defitelio), cardiology (Irbesartan), dermatology (Voluma, Botox), neuroscience (Abilify) and anti-infectives (Teflaro). Since JuneNovember 2018, Dr. Smith has been providing consulting services internationally. Dr. Smith currently serves as Chief Medical Officer for Emergent BioSolutions, Inc., a public biopharmaceutical company. From May 2019 to January 2020, Dr. Smith served as President and Chief Executive Officer of Medeor Therapeutics, Inc., a biotechnology company. From June 2018 to May 2019, Dr. Smith served as Chief Executive Officer of Eliminate Cancer, Inc. From April 2015 to May 2018, she served as the Global Head of Research & Development and Chief Medical Officer of Jazz Pharmaceuticals plc.plc, a biopharmaceutical company, where she built the R&D function into a pipeline of neuroscience and oncology products across all stages of discovery and development. From January 2011 to March 2015, she was Senior Vice President, Global Medical Affairs and Global Therapeutic Area Head (Dermatology) for Allergan, Inc., a multi-specialty health care company. Earlier in her career, she held senior leadership roles at AstraZeneca plc and Bristol Myers Squibb Company. Dr. Smith received a B.App.Sci in biotechnology and a B.Sc. in haematology from Curtin University of Technology,holds several degrees, including an M.D. from the University of Warwick, a Ph.D. in oncology molecular genetics from the University of Western Australia, an M.B.A. from the University of New England (Australia) and an L.L.M. (Masters in medical lawLaw) from the University of Salford. Dr. Smith has served on the Boards of Forward Pharma and Sucampo Pharmaceuticals and currently serves on the boardsboard of directors of Acceleron Pharma, Inc., a public biopharmaceutical company, and Antares Pharma, Inc., a public pharmaceutical company. Dr. Smith previously served on the
board of directors of Sucampo Pharmaceuticals, Inc. from July 2017 to February 2018, and Forward Pharma A/S, from June 2016 to June 2017, and serves as the chair of the Strategic Advisory Board of Emyria Limited, a healthcare technology and services company. The Nominating and Corporate Governance Committee and the Board believe that Dr. Smith’s extensive executive experience in global research and development, combined with tenure on prior public company boards provides us with access to a valuable
skill-set as we translate our science into genomic medicines using our platform technologies in gene editing, gene therapy, gene regulation and cell therapy.
In addition, the Nominating and Corporate Governance Committee and the Board do not believe that Dr. Smith’s outside boards or other commitments limit her ability to devote sufficient time and attention to her duties as a director of Sangamo, particularly given her exemplary attendance record at meetings of the Board and of the Compensation Committee.
Joseph S. Zakrzewski, age 56,58, has served as a member of our Board of Directors since June 2017. Mr. Zakrzewski has over 25 years of experience in the biopharmaceutical industry with senior leadership experience in R&D, supply chain and manufacturing operations, business development and commercialization.9
Prior to focusing on serving on boards and investments, from 2010 through 2013, he was Chairman and Chief Executive Officer of Amarin Corporation PLCplc (formerly Amarin Pharmaceuticals, Inc.), where he led the development and commercialization of the company’s first product, Vascepa. Mr. Zakrzewski previously served as a Venture Partner with Orbimed Advisors LLC, a venture capital firm, and as Chairman, President and Chief Executive Officer of Xcellerex, Inc., a privately held company focused on the commercialization of its proprietary manufacturing technology for biotherapeutics and vaccines. Earlier, he served as Chief Operating Officer of Reliant Pharmaceuticals, Inc. before its acquisition by GlaxoSmithKline in 2007 and held various executive positions at Eli Lilly & Company in the areas of R&D, manufacturing, finance and business development. Mr. Zakrzewski currently serves as a member ofon the board of directors of various public and private companies, including Acceleron Pharma, Inc., a public biopharmaceutical company, and Amarin and Onxeo SA where he serves as Chairman.Corporation plc, a public biopharmaceutical company. Mr. Zakrzewski previously served on the board of directors of Insulet Corporation, a public medical device company, from 2008 to August 2017. Mr. Zakrzewski received ahis B.S. in Chemical Engineering and an M.S. in Biochemical Engineering from Drexel University, and an M.B.A. in Finance from Indiana University. The Nominating and Corporate Governance Committee and the Board believe that Mr. Zakrzewski’s significant experience as a Chief Executive Officer and leading a variety of functional areas including supply and manufacturing operations provides the Board with an important set of skills to assist in the oversight of the development of our novel genomic and cell therapy assets.
Recommendation of the Board of Directors
RECOMMENDATION OF THE BOARD OF DIRECTORS
The Board of Directors recommends that the stockholders vote FOR the election of each the nominees listed above.
Stockholder Engagement and Responsiveness
A priority for our Board of Directors is soliciting and listening to the views of our stockholders on a variety of topics, including our business and growth strategy, corporate governance practices and executive compensation matters.
In this regard, we regularly engage with our institutional investors, and since our 2018 annual meeting of stockholders, we reached out to investors representing over 25% of our outstanding shares. Our discussions with our investors have been productive and informative and have provided valuable feedback to our Board of Directors to help ensure that our Board’s decisions are aligned with stockholder objectives.
Our Board of Directors is also committed to being responsive to the concerns of our stockholders. At our 2018 annual meeting of stockholders, Dr. Roger Jeffs was elected to the Board at the 2018 annual meeting of stockholders with the support of approximately 71.5% of the votes cast in favor of his election. While this was an overall strong expression of support, it was lower than the level of support received by our other nominees in 2018 and as a result, we determined that further stockholder engagement was necessary to determine the reason for such result. Through a combination of our outreach to the institutional investors who we believe did not vote in favor of Dr. Jeffs’ election and our review of the published voting guidelines and policies of those institutional investors, we believe that the lower level of support for Dr. Jeffs’ election vis-à-vis our other nominees was primarily due to concerns about “overboarding,” which refers to the situation where a director serves on more than a prescribed number of public company boards.
In April 2019, Dr. Jeffs notified us of his decision to not stand for re-election at the Annual Meeting and will be resigning from the Board effective as of the Annual Meeting. Dr. Jeffs indicated to us that his decision not to stand for re-election and his resignation from the Board was due to his other professional commitments, including his other public company directorships.
Our Board of Directors is also focused on overboarding concerns and strongly believes that all members of our Board of Directors should have sufficient time and attention to devote to Board duties and to otherwise fulfill the responsibilities required of directors. Prior to recommending a candidate as a nominee for director, the Nominating and Corporate Governance Committee reviews the candidate’s professional commitments, including the number of boards on which the candidate serves and considers whether those commitments may limit the ability of the candidate to devote sufficient time and attention to Board duties. Although Dr. Jeffs was deemed
10
overboarded based on the criteria adopted by certain of our institutional investors, neither the Nominating and Corporate Governance Committee nor our Board of Directors believed that Dr. Jeffs’ outside board or other professional commitments ever limited his ability to devote sufficient time and attention to Board duties.
The Board of Directors has determined that each of its
current and nominated directors is independent under applicable listing
standardstandards of
The Nasdaq Stock Market LLC, or Nasdaq, except for Dr. Macrae, who is our Chief Executive Officer. There are no family relationships between any of our directors and any of our executive officers.
Board Committees and Meetings
The Board of Directors held
eightseven meetings
and acted once by unanimous written consent in lieu of a meeting during
2018.2020. The Board of Directors has an Audit Committee, a Compensation Committee and a Nominating and Corporate Governance Committee. Each director attended or participated in 75% or more of the aggregate of (i) the total number of meetings of the Board of Directors and (ii) the total number of meetings held by all committees of the Board on which such director served during
2018,2020, in each case held during the period for which each respective director was serving as a director.
The Audit Committee consists of three directors: Mr. Carey, Mr. Meyers and Ms. Ramasastry,Ramasastry. Mr. Meyers was appointed to serve on the Audit Committee effective September 30, 2020 to replace Dr. Stephen G. Dilly, a former member of the Board who resigned from the Board and Mr. Zakrzewski.Audit Committee effective September 30, 2020. Mr. Carey serves as the ChairmanChair of this committee. Our Board of Directors has determined that each member of the Audit Committee is independent under the applicable listing standard standards of Nasdaq and SEC rules, and that Dr, Dilly was independent under the applicable listing standards
of Nasdaq and SEC rules. The Board of Directors has determined that Mr. Carey is an “audit committee financial expert” as defined under SEC rules and
that each member of the Audit Committee has the requisite financial sophistication in accordance with the applicable Nasdaq listing standards. The Audit Committee held four meetings during
2018.2020.
The Audit
Committee assistsCommittee’s primary purposes are to (i) assist the Board
of Directors in its oversight of the integrity of our financial statements,
our systems of accounting and financial controls, our accounting and financial reporting processes and the
risk management andaudit of our
internal controls and our compliance with legal and regulatory requirements. The Audit Committee interactsfinancial statements, (ii) interact directly with and
evaluatesevaluate the performance of
theour independent
registered public accounting firm, including determiningauditors, determine whether to engage or dismiss
theour independent
registered public accounting firmauditors and
to monitor
theour independent
registered public accounting firm’sauditors’ qualifications and
independence.independence and (iii) oversee the implementation of our compliance program with respect to financial, accounting, auditing, information technology and cybersecurity matters. The Audit Committee also
pre-approves all audit services and permissible
non-audit services provided by
theour independent
registered public accounting firm.auditors. The Audit Committee Report is included herein
on page 60.in the section labeled “Report of the Audit Committee of the Board of Directors.” The Audit Committee has a written charter, which is available on our website at https://investor.sangamo.com/corporate-governance/governance-overview.
The Compensation Committee consists of three directors:
Drs. MentoMr. Meyers, Dr. Smith and
Jeffs and Ms. Parker, each of whom is independent under applicable Nasdaq and SEC rules. Dr. Mento serves as Chairman of this committee. After the conclusion of the Annual Meeting, the Compensation Committee will be reconstituted to reflect Drs. Mento’s and Jeffs’ departures from the Board, including all of its committees. The Board hasMr. Zakrzewski. Mr. Meyers was appointed
effective at the time of the completion of the Annual Meeting, each of Mr. Zakrzewski and Dr. Smith to serve on the Compensation Committee
witheffective April 1, 2020. Ms. Parker previously served on the Compensation Committee through March 2020. Mr. Zakrzewski
servingserves as
ChairmanChair of this committee.
Our Board of Directors has determined that each member of the Compensation Committee is independent under the applicable listing standards of Nasdaq and SEC rules.
The Compensation Committee’s responsibilities include,
among other things (i)
approving a philosophy for compensation of our executive officers and key employees; (ii) adopting, administering and reviewing compensation
and benefit plans and programs for our executive officers
and key employees, including
incentiveequity and
equity plansretirement plans; (iii) approving executive employment contracts and
programs; (ii) approving compensation arrangements for our executive officers; (iii) evaluating the performance of our executive officers;severance and change in control arrangements; (iv)
adjusting compensation arrangements as appropriate based upon performance;11
(v) reviewing, considering and approving compensation programs applicable tonon-employee members of the BoardBoard; (v) approving general compensation levels for our executive officers, with the exception of Directors;Dr. Macrae, and key employees; (vi) evaluating the performance and leadership of Dr. Macrae and determining his total compensation package; (vii) reviewing with Dr. Macrae his evaluation of the performance of the executive officers other than him and of our key employees, and approving each such executive officer’s and key employee’s total compensation package; (viii) reviewing and monitoring management development and succession plans and activities and (vii) administering, concurrentlysuccession plans; (ix) conducting risk assessments of our compensation programs; (x) considering stock ownership guidelines for directors and employees; and (xi) reviewing and discussing with the Board of Directors, the Sangamo Therapeutics Inc. 401(k) Profit Sharing Planmanagement our Compensation Discussion and Trust, originally effectiveAnalysis and preparing a report on January 1, 1998, and as most recently amended and restated as of June 24, 2015, or the 401(k) Plan.
executive compensation, each for inclusion in this Proxy Statement.
The Compensation Committee may delegate any responsibility or authority of the Compensation Committee under its charter to one or more members of the Compensation Committee, as appropriate and as consistent with applicable laws and rules.
Further, the Compensation Committee may delegate to one or more employees or one or more other members of the Board, the authority to grant awards of equity securities pursuant to our equity incentive plans, in each case to any persons who are not executive officers or key employees in accordance with such guidelines as may be established by the Compensation Committee. The Compensation Committee
may also form anddoes not, however, delegate
authorityany of its functions to
a committee composed solely of our employees to serve as an administrative and/others in determining or
investment committee, with fiduciary responsibilities under the Employee Retirement Income Security Act of 1974,recommending director or
ERISA, with respect to one or more of our plans that are subject to ERISA. The Compensation Committee may also form and delegate authority to a subcommittee composed solely of at least two members of the Compensation Committee who are“non-employee directors” under Rule16b-3 to grant awards of equity securities and to take such other actions as may be necessary or appropriate to qualify transactions in our equity securities under the Rule16b-3 exemptions.executive officer compensation.
The Compensation Committee is authorized to engage, oversee and terminate independent compensation consultants and other professionals to assist in the design, formulation, analysis and implementation of compensation programs for our executive officers and other key employees. The Compensation Committee retained the services of Radford,
awhich is part
of the Rewards Solutions practice of Aon plc, or Radford, in order to (i) assess compensation levels and mix of elements for our executive officers and vice presidents for
2018,2020, (ii) review the peer group criteria and to recommend specific companies, (iii) assess the compensation of the
non-employee directors and (iv) advise the
committeeCompensation Committee on executive compensation and governance trends based on peer group trends and market practices.
The Compensation Committee held
ninesix meetings
and acted once by unanimous written consent in lieu of a meeting during
2018.2020. The Compensation Committee has a written charter, which is available on our website at https://investor.sangamo.com/corporate-governance/governance-overview. For information regarding our processes and procedures for the consideration and determination of executive and director compensation, please see “Executive Compensation—Compensation Discussion and Analysis” and “—Director Compensation,” respectively.
Compensation Committee Interlocks and Insider Participation
During
2018, Drs. Mento and Jeffs and2020, Mr. Meyers, Ms. Parker,
Dr. Smith and Mr. Zakrzewski served on the Compensation Committee. None of our Compensation Committee members has been an officer or employee of Sangamo at any time. None of our executive officers serves on the board of directors or compensation committee of any entity that has one or more executive officers serving as a member of our Board or our Compensation Committee.
Nominating and Corporate Governance Committee
The Nominating and Corporate Governance Committee consists of three directors: Dr. Dilly, Ms. Parker andHillan, Dr. Jeffs, each of whom is independent under applicable Nasdaq listing standards. Dr. Jeffs serves as the Chairman of the Nominating and Corporate Governance Committee. After the conclusion of the Annual Meeting, the Nominating and Corporate Governance Committee will be reconstituted to reflect Dr. Jeffs’ departure from the Board, including all of its committees. The Board has appointed, effective at the time of the completion of the Annual Meeting, each of Dr. DillyMarkels and Ms. ParkerParker. Dr. Markels was appointed to serve on the Nominating and Corporate Governance Committee. Committee effective April 1, 2020. Dr. Hillan was appointed to serve on the Nominating and Corporate Governance Committee effective September 30, 2020. Mr. Meyers previously served on the Nominating and Corporate Governance Committee through March 2020, and Dr. Dilly previously served on the Nominating and Corporate Governance Committee until his resignation from our Board of Directors effective September 30, 2020. Ms. Parker serves as Chair of this committee. Our Board of Directors has determined that each member of the Nominating and Corporate Governance Committee is independent under the applicable listing standards of Nasdaq and SEC rules.
The Nominating and Corporate Governance Committee considers and periodically reports on matters relating to the size, identification, selection and qualification of the Board of Directors and candidates nominated
12
for the Board of Directors and its committees, and develops and recommends governance principles and policies applicable to the Company.
It also assists the Board in its oversight of our compliance with legal and regulatory requirements relating to matters other than financial, accounting, auditing, information technology and cybersecurity matters.
The Nominating and Corporate Governance Committee held
twofour meetings
and acted once by unanimous written consent in lieu of a meeting during
2018.2020. The Nominating and Corporate Governance Committee has a written charter, which is available on our website at https://investor.sangamo.com/corporate-governance/governance-overview. The Nominating and Corporate Governance Committee considers properly submitted stockholder recommendations for candidates for membership on the Board of Directors as described below under “—Identification and Evaluation of Nominees for Directors.” In evaluating such recommendations, the Nominating and Corporate Governance Committee seeks to achieve a balance of knowledge, experience and capability on the Board of Directors and to address the membership criteria set forth below under “—Director
Qualifications.Qualifications & Composition.”
Director Qualifications
& Composition
The Nominating and Corporate Governance Committee will use a variety of criteria to evaluate the qualifications and skills necessary for members of our Board of Directors. The Nominating and Corporate Governance Committee may assess character, judgment, business acumen and scientific expertise, and familiarity with issues affecting the biotechnology and pharmaceutical industries. Other qualifications will be determined on a
case-by-case basis, depending on whether the Nominating and Corporate Governance Committee desires to fill a vacant seat or increase the size of the Board to add new directors. In addition, the Nominating and Corporate Governance Committee may also evaluate whether a potential director nominee’s skills are complementary to existing Board members’ skills or meet the Board’s need for operations, management, commercial, financial or other expertise.
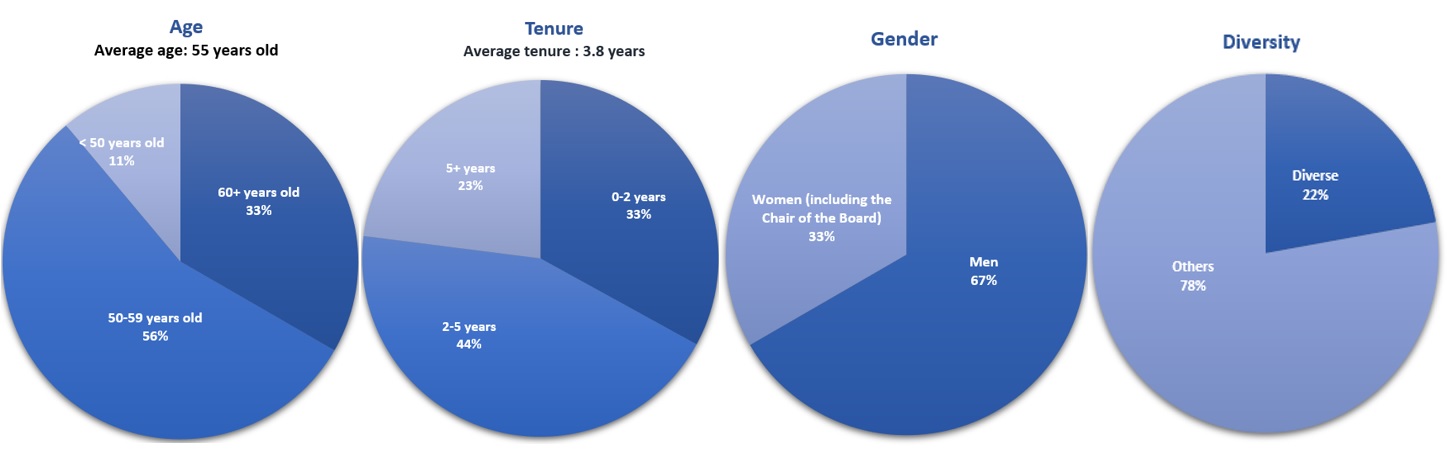
While theThe Nominating and Corporate Governance Committee
does not prescribe specificstrives to maintain a diverse Board reflecting a variety of skills, experiences, perspectives and backgrounds, including age, tenure, gender, race, ethnicity, sexual orientation and other unique characteristics, as it believes that such diversity
standards, as a matterenhances the Board’s effectiveness in fulfilling its oversight role. Below we highlight certain attributes of
practice, the committee considers diversity in the contextour current directors. For purposes of the
Boardinformation below, a ‘diverse’ director is a member of an underrepresented community under California AB 979, defined as
a whole and takes into account the personal characteristics and experiences of current and prospective directors that reflect a broad range of perspectives in the Board’s decision-making process.‘an individual who self-identifies as Black, African American, Hispanic, Latino, Asian, Pacific Islander, Native American, Native Hawaiian, or Alaska Native, or who self-identifies as gay, lesbian, bisexual or transgender.’
Identification and Evaluation of Nominees for Directors
The Nominating and Corporate Governance Committee utilizes a variety of methods for identifying and evaluating nominees for director. The Nominating and Corporate Governance Committee assesses the appropriate size of the Board of Directors, and whether any vacancies on the Board of Directors are expected due to retirement or otherwise. In the event that
vacancies are anticipated, or otherwise arise, the Nominating and Corporate Governance Committee considers various potential candidates for director. Candidates may come to the attention of the Nominating and Corporate Governance Committee through current members of the Board of Directors or senior management, executive recruiting firms, stockholders or other persons.
TheseFor example, Dr. Hillan was initially recommended to the Nominating and Corporate Governance Committee by our Chief Executive Officer upon the recommendation of another Board Director. In addition, we have in the past and may from time to time again in the future engage a third-party search firm to assist in identifying potential directors. The Board often considers specific industry expertise such as research, development or commercial experience in candidates
to make sure the Board is able to oversee all aspects of our business. Candidates for director are evaluated at regular or special meetings of the Nominating and Corporate Governance Committee and may be considered at any point during the year. The Nominating and Corporate Governance Committee
considerswill consider properly submitted stockholder recommendations for candidates for the Board of Directors.
Stockholder nomineesNominees recommended by stockholders will receive the same consideration that nominees of the Board receive. Any stockholder recommendations proposed for consideration by the Nominating and Corporate Governance Committee must provide all information requested by the Nominating and Corporate Governance Committee relating to such recommendation, including the candidate’s name and qualifications for membership on the Board of Directors and should be addressed to Investor Relations at the following address:
Investor Relations Department
Sangamo Therapeutics, Inc.
501 Canal
7000 Marina Boulevard
Richmond,
Brisbane, CA
9480413
94005
In evaluating such recommendations, the Nominating and Corporate Governance Committee applies the qualifications standards discussed above and seeks to achieve a balance of knowledge, experience and capability on the Board of Directors.
Leadership Structure of the Board
Under our Bylaws, the Board is not required to appoint our Chief Executive Officer as the Chair of the Board, and the Board does not have a policy on whether or not the roles of Chief Executive Officer and Chair of the Board should be separate. Currently two individuals serve in these two positions. Ms. Parker currently serves as the Chair of the Board. Ms. Parker has extensive knowledge and experience in the life science industry and an
in-depth understanding of our business strategies and
day-to-day operations, which makes her well suited to set the agenda and lead the discussions at Board meetings as the Chair. The Chair is responsible for chairing Board meetings and meetings of stockholders, setting the agenda for Board meetings and providing information to the Board members in advance of meetings and between meetings. In addition, our Chief Executive Officer, Dr. Macrae, also serves as a director on our Board. The Board believes that Dr. Macrae’s membership as a director provides the Board with an
in-depth understanding of our business operations because of his extensive
experiencesexperience and knowledge of the
day-to-day management of all aspects of our operations. This also facilitates communications between the Board and management by ensuring a regular flow of information, thereby enhancing the Board’s ability to make informed decisions on critical issues facing our company.
Of the
sevennine directors on the Board following the Annual Meeting,
sixeight directors are independent under applicable Nasdaq corporate governance rules. The Board believes that this establishes a strong independent board that provides effective oversight of the Company. Moreover, in addition to feedback provided during the course of Board meetings, the independent directors conduct regular executive sessions without the presence of Dr. Macrae or any other members of management. We believe that our leadership structure of the Board is appropriate given the nature and size of our business, because it provides both effective independent oversight and expertise in the complexity and management of our operations as a life sciences company.
Oversight of Risk Management by the Board
Our Board of Directors is generally responsible for the oversight of corporate risk in its review and deliberations relating to our
activities and has determined that our principal source of risk falls into two categories: (1) financial and (2) product development and testing.activities. The Audit Committee oversees management of risks associated with our financial and accounting systems, public financial reporting, investment strategies and policy, and certain other matters delegated to the Audit Committee, including risks associated with our information technology systems (including cybersecurity risks). Our Board of Directors regularly reviews information regarding our cash position, liquidity and operations, as well as the risks associated with each. The Board regularly reviews plans, results and potential risks related to our lead therapeutic development programs and other preclinical programs as well as financial and strategic
riskrisks related to our
business and operations.
In addition, the Nominating and Corporate Governance Committee monitors the effectiveness of our corporate policies and manages risks associated with the independence of the Board of Directors and potential conflicts of interest. Our Compensation Committee oversees risk management as it relates to our compensation plans, policies and practices for all employees, including executives, particularly whether our compensation programs may create incentives for our employees to take excessive or inappropriate risks that could have a material adverse effect on the Company. While each committee is
responsible for evaluating certain risks and overseeing the management of such risks, the entire Board of Directors is
regularlyperiodically informed
through committee reports about such risks.
In May 2020, upon the request of the Board, we established an internal compliance committee staffed by employees led by our General Counsel and Chief Compliance Officer, who reports directly to the Board on compliance matters. The compliance committee held quarterly meetings in 2020, starting with the second quarter. We also established a COVID-19 Taskforce in 2020 responsible for developing our internal COVID-19 policies and protocols. This Taskforce meets regularly and was led by our former Chief Financial Officer in 2020 and is now led by our Chief People Officer.
Annual Meeting Attendance
Although we do not have a formal policy regarding attendance by members of the Board of Directors at our annual meetings of stockholders,
we encourage our directors
are encouraged to attend annual meetings of our stockholders. All of our then current directors
who were nominated for re-election attended the
20182020 annual meeting of
stockholders.14
stockholders, which was held online-only in light of public health concerns regarding the ongoing COVID-19 pandemic.
Communications with the Board of Directors
Our Board of Directors currently does not have a formal process for stockholders to send communications to the Board of Directors. Although we do not have a formal policy regarding communications with the Board of Directors, stockholders may communicate with the Board of Directors, including the
non-management directors, by sending a letter to the Sangamo Board of Directors, c/o Investor Relations,
501 Canal7000 Marina Boulevard,
Richmond,Brisbane, California
94804.94005. Stockholders who would like their submission directed to a particular member of the Board of Directors may so specify.
The Board of Directors does not recommend that formal communication procedures be adopted at this time because it believes that informal communications are sufficient to communicate questions, comments and observations that could be useful to the Board.
Code of Business Conduct and Ethics
The Board of Directors has adopted a Code of Business Conduct and Ethics, which is applicable to all employees,
including our executive officers, and directors of the Company. A copy of our Code of Business Conduct and Ethics is available on our website at https://investor.sangamo.com/corporate-governance/governance-overview in the Investors
&+ Media Section under Corporate Governance. In the event that we make any amendments to or grant any waivers of, a provision of the Code of Business Conduct and Ethics that
applies to the principal executive officer, principal financial officer, or principal accounting officer that requires disclosure under applicable SEC rules, we intend to disclose such amendment or waiver and the reasons
therefore,therefor, on our website.
Prohibitions on Hedging, Pledging and Speculative Transactions
Under the terms of our insider trading policy, none of our directors, officers and other employees may engage in any hedging or monetization transactions relating to our securities, including through the use of financial instruments such as prepaid variable forwards, equity swaps, collars and exchange funds. In addition, all of our officers, directors and employees are prohibited from short-selling our securities or engaging in transactions involving Sangamo-based derivative securities (other than those granted under our employee stock option or equity incentive plans) and are further prohibited from holding our securities in a margin account or otherwise pledging our securities as collateral for a loan.
The following table sets forth certain information regarding the compensation of each
non-employee director for service as a member of the Board of Directors during
2018. | | | | | | | | | | | | | | | | |
Name | | Fees Earned or
Paid in Cash
($) (1) | | | Option Awards
($) (2) (3) (5) (6) | | | Stock Awards
($) (4) (5) | | | Total
($) | |
Robert F. Carey | | | 60,000 | | | | 151,145 | | | | 39,125 | | | | 250,270 | |
Stephen G. Dilly | | | 45,000 | | | | 151,145 | | | | 39,125 | | | | 235,270 | |
Roger Jeffs (7) | | | 55,361 | | | | 151,145 | | | | 39,125 | | | | 245,631 | |
Steven J. Mento (7) | | | 55,000 | | | | 151,145 | | | | 39,125 | | | | 245,270 | |
H. Stewart Parker | | | 87,500 | | | | 151,145 | | | | 39,125 | | | | 277,770 | |
Saira Ramasastry | | | 50,000 | | | | 151,145 | | | | 39,125 | | | | 240,270 | |
Karen L. Smith (8) | | | 20,000 | | | | 302,289 | | | | 78,250 | | | | 400,539 | |
Joseph S. Zakrzewski | | | 50,000 | | | | 151,145 | | | | 39,125 | | | | 240,270 | |
(1) | Consists of the annual retainer fee for service as a member of the Board of Directors or any Board committee. For further information concerning such fees, see the section below entitled “—Director Annual Retainer and Meeting Fees.”
|
(2) | Represents the grant date fair value of the awards computed in accordance with Financial Accounting Standards Board, or FASB, Accounting Standards Codification Topic 718,Compensation—Stock Compensation, or ASC 718.
|
(3) | Pursuant to the automatic grant program in effect under our 2018 Equity Incentive Plan, or the 2018 Plan, Mr. Carey, Dr. Dilly, Dr. Jeffs, Dr. Mento, Ms. Parker, Ms. Ramasastry, and Mr. Zakrezewski each received an option to purchase 15,000 shares of common stock with an exercise price per share of $15.65 on the date of the 2018 annual meeting of stockholders, and each such option had an aggregate grant date fair value of $151,145. On June 26, 2018, Dr. Smith received an option to purchase 30,000 shares of common stock with an exercise price per share of $15.65 in connection with her appointment to the Board of Directors on June 26, 2018 and such option had an aggregate grant date fair value of $302,289.
|
(4) | Pursuant to the automatic grant program in effect under the 2018 Plan, Mr. Carey, Dr. Dilly, Dr. Jeffs, Dr. Mento, Ms. Parker, Ms. Ramasastry, and Mr. Zakrezewski each received an award of 2,500 restricted stock units, or RSUs, on the date of the 2018 annual meeting of stockholders. On June 26, 2018, Dr. Smith received an award of 5,000 RSUs in connection with her appointment to the Board of Directors.
|
15
2020. Dr. Macrae, our President and Chief Executive Officer, is not listed in the following table because he is our employee. Dr. Macrae’s compensation is described under “Executive Compensation” and he received no additional compensation for serving on our Board of Directors in 2020.
(5) | Aggregate grant date fair values are calculated in accordance with ASC 718 using the Black-Scholes option valuation model and without taking into account any estimated forfeitures. The assumptions used in the calculation of such grant date fair values are described in Note 4 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the year ended December 31, 2018 filed with the SEC on March 1, 2019.
|
(6) | As of December 31, 2018, the followingnon-employee directors held options to purchase the following number of shares of our common stock: Mr. Carey, 55,000 shares; Dr. Dilly, 135,000 shares; Mr. Jeffs, 45,000 shares; Dr. Mento, 55,000 shares; Ms. Parker, 95,000 shares; Ms. Ramasastry, 65,000 shares; Dr. Smith, 30,000 shares; and Mr. Zakrzewski 45,000 shares.
|
(7) | Drs. Mento and Jeffs are not standing for reelection to the Board of Directors at the Annual Meeting.
|
(8) | Dr. Smith was appointed as anon-employee member of the Board of Directors on June 26, 2018; accordingly, her retainer fees werepro-rated over her period of service in 2018.
|
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | | Fees Earned or
Paid in Cash
($) (1) | | Option Awards
($) (2) (3) (5) | | Stock Awards
($) (2) (4) (6) | | Total
($) |
| Robert F. Carey | | 60,000 | | | 135,572 | | | 104,600 | | | 300,172 | |
| Stephen G. Dilly (7) | | 45,000 | | | 135,572 | | | 104,600 | | | 285,172 | |
| Kenneth J. Hillan (8) | | 13,655 | | | 200,988 | | | 156,450 | | | 371,093 | |
| John H. Markels (9) | | 43,750 | | | 147,900 | | | 38,000 | | | 229,650 | |
| James R. Meyers | | 49,402 | | | 135,572 | | | 104,600 | | | 289,574 | |
| H. Stewart Parker | | 83,152 | | | 135,572 | | | 104,600 | | | 323,324 | |
| Saira Ramasastry | | 50,000 | | | 135,572 | | | 104,600 | | | 290,172 | |
| Karen L. Smith | | 47,500 | | | 135,572 | | | 104,600 | | | 287,672 | |
| Joseph S. Zakrzewski | | 55,000 | | | 135,572 | | | 104,600 | | | 295,172 | |
___________________
(1)Consists of the annual retainer fee for service as a member of the Board of Directors or any Board committee. For further information concerning such fees, see the section below entitled “—Director Annual Retainer and Meeting Fees.”
(2)Represents the grant date fair value of the awards computed in accordance with Financial Accounting Standards Board Accounting Standards Codification Topic 718, Compensation—Stock Compensation, or ASC 718. The assumptions used in the calculation of such grant date fair values are described in Note 9 of the Notes to Consolidated Financial Statements included in our Annual Report on Form 10-K for the year ended December 31, 2020 filed with the SEC on February 24, 2021, or the 2020 Form 10-K.
(3)Pursuant to the automatic grant program under our Amended and Restated 2018 Equity Incentive Plan, or the 2018 Plan, Mr. Carey, Dr. Dilly, Mr. Meyers, Ms. Parker, Ms. Ramasastry, Dr. Smith and Mr. Zakrzewski each received an option to purchase 20,000 shares of common stock with an exercise price per share of $10.46 on the date of the 2020 annual meeting of stockholders, and each such option had an aggregate grant date fair value of $135,572. On February 11, 2020, Dr. Markels received an option to purchase 30,000 shares of common stock with an exercise price per share of $7.60 in connection with his appointment to the Board of Directors and such option had an aggregate grant date fair value of $147,900. On September 9, 2020, Dr. Hillan received an option to purchase 30,000 shares of common stock with an exercise price per share of $10.43 in connection with his appointment to the Board of Directors and such option had an aggregate grant date fair value of $200,988.
(4)Pursuant to the automatic grant program under the 2018 Plan, Mr. Carey, Dr. Dilly, Mr. Meyers, Ms. Parker, Ms. Ramasastry, Dr. Smith and Mr. Zakrzewski each received an award of 10,000 restricted stock units, or RSUs, on the date of the 2020 annual meeting of stockholders and each such RSU award had an aggregate grant date fair value of $104,600. On February 11, 2020, Dr. Markels received an award of 5,000 RSUs in connection with his appointment to the Board of Directors and such RSU award had an aggregate grant date fair value of $38,000. On September 9, 2020, Dr. Hillan received an award of 15,000 RSUs in connection with his appointment to the Board of Directors and such RSU award had an aggregate grant date fair value of $156,450.
(5)As of December 31, 2020, the following non-employee directors held options to purchase the following number of shares of our common stock: Mr. Carey, 90,000 shares; Dr. Hillan, 30,000 shares; Dr. Markels, 30,000 shares; Mr. Meyers, 50,000 shares; Ms. Parker, 130,000 shares; Ms. Ramasastry, 90,000 shares; Dr. Smith, 65,000 shares; and Mr. Zakrzewski, 80,000 shares.
(6)As of December 31, 2020, the following non-employee directors held RSUs for the following number of shares of our common stock: Mr. Carey, 10,000 shares; Dr. Hillan, 15,000 shares; Dr. Markels, 5,000 shares; Mr. Meyers, 13,333 shares; Ms. Parker, 10,000 shares; Ms. Ramasastry, 10,000 shares; Dr. Smith, 11,667 shares; and Mr. Zakrzewski, 10,000 shares.
(7)Dr. Dilly resigned from the Board of Directors effective September 30, 2020; accordingly, his retainer fees were prorated over his period of service in 2020.
(8)Dr. Hillan was appointed as a non-employee member of the Board of Directors on September 9, 2020; accordingly, his retainer fees were prorated over his period of service in 2020.
(9)Dr. Markels was appointed as a non-employee member of the Board of Directors on February 11, 2020; accordingly, his retainer fees were prorated over his period of service in 2020.
Processes and Procedures for Determining Director
Compensation.Compensation
The charter of the Compensation Committee provides the Compensation Committee with the responsibility for reviewing, considering and approving compensation programs applicable to
non-employee directors. It is the practice of the Compensation Committee to seek input from outside compensation consultants, including Radford, our Compensation Committee’s compensation consultant, as it deems appropriate.
Director Annual Retainer and Meeting Fees
Eachnon-employee member of the Board of Directors receives an annual cash retainer of $40,000. To the extent the Board of Directors meets more than 10 times in any year, eachnon-employee member of the Board of Directors will receive, for each meeting in excess of 10, a per meeting fee of $1,000 if attended in person and $500 if attended by video or telephone conference. The Chair of the Board of Directors receives an additional annual cash retainer of $35,000. In addition, thenon-employee members of the Board of Directors serving as the ChairmanChair of the Audit Committee, the ChairmanChair of the Compensation Committee and the ChairmanChair of the Nominating and Corporate Governance Committee receive an additional annual cash retainer of $20,000,
$20,000, $15,000 and $10,000, respectively. Each
non-employee member of the Board of Directors serving as a member of the Audit Committee, the Compensation Committee and the Nominating and Corporate Governance Committee, other than the
Chairman,Chair, receives an additional annual cash retainer of $10,000, $7,500 and $5,000, respectively. All retainer fees are subject to
pro-ration proration for directors who join or leave the Board of Directors or the respective committee or begin or cease to serve as Chair of the Board or the respective committee, during the year.
Equity Incentives Granted Under
the 2018
Stock Incentive Plan
Under the automatic grant program
underof the 2018 Plan,
on the date of each annual stockholders meeting, each individual
serving aswho is a
non-employee Board member
at that time, who has served on our Board for at least six months, and who will continue serving as anon-employee Board member immediately after such stockholders meeting will automatically be granted a nonqualified stock option, or NSO, to purchase
15,000common stock and a RSU award on an annual basis as follows: (i) with respect to 2020, a NSO to purchase 20,000 shares of common stock and a RSU
in respectaward of
2,50010,000 shares of common stock
underwere granted on the
2018 Plan.date of the 2020 Annual Meeting; and (ii) with respect to each year following 2020, a NSO grant and a RSU award will be granted on the 25th day of February of such year (or if such 25th day is not a trading day, the immediately preceding trading day in February); in each case, provided that such individual is a non-employee Board member on the date of grant, has served as a non-employee Board member for at least three months prior to the date of grant, and will continue serving as a non-employee Board member immediately after the date of grant. The number of shares to be automatically granted annually pursuant to a NSO or a RSU award are determined based on a review of our share price and comparative market data from peer companies. For 2021, a NSO grant of 16,400 shares and a RSU award of 8,200 shares of common stock were granted on February 25, 2021. The shares subject to each annual
15,000 share automatic NSO
grant madegranted to a continuing Board member will vest in 12 successive equal monthly installments
upon suchover the one-year period following the date of grant, subject to the director’s
completion of each month of Boardcontinuous service
over the12-month period measured fromthrough the
grant date.applicable vesting dates. The shares subject to
each 2,500 sharethe 2020 automatic RSU
grantaward granted to a continuing Board member in 2020 will fully vest on the earlier of (x) the first anniversary of the date of grant or (y) the day prior to the next annual
stockholdersstockholders’ meeting, subject to the director’s
continuedcontinuous service
on the Board through the applicable vesting date.
The shares subject to each annual automatic RSU granted to a continuing Board member following 2020 will fully vest on the first anniversary of the date of grant, subject to the director’s continuous service through the applicable vesting date.
Under the automatic grant program
underof the 2018 Plan, each
non-employee Board member individual who is initially appointed or elected
toas a non-employee Board member on or after the
Board2020 Annual Meeting will,
on the date of such appointment or election, automatically be granted
a NSO to purchase common stock and a RSU award on an
annual basis as follows: (i) with respect to 2020, a NSO to purchase 30,000 shares of common
16
stock and a RSU award inof 15,000 shares of common stock; and (ii) with respect of 5,000to 2021, a NSO to purchase 24,600 shares of common stock under the 2018 Plan.and a RSU award of 12,300 shares of common stock. The number of shares to be automatically granted pursuant to an initial NSO or a RSU award are determined based on a review of our share price and comparative market data from peer companies. The shares subject to each initial 30,000 share automatic NSO grant madegranted to a newly appointed or elected Board member will vest in 36 successive equal monthly installments upon suchover the three-year period following the date of grant, subject to the director’s completion of each month of Boardcontinuous service overthrough the36-month period measured from the grant date. applicable vesting dates. The shares subject to each 5,000 shareinitial automatic RSU grantaward granted to a newly appointed or elected Board member will vest with respect to 1/3rd of the shares in three equal annual installments over the three-year period measured fromfollowing the date of grant.
grant, subject to the director’s continuous service through the applicable vesting dates.
Each automatic NSO grant will have an exercise price per share equal to the fair market value per share of our common stock on the grant date and will have a term of 10 years, subject to earlier termination following the optionee’s cessationdirector’s termination of Boardcontinuous service. Each automatic NSO option grant will be immediately exercisable for all of the option shares; however, we may repurchase, at the lower of the exercise price paid per share or the fair market value per share, any shares purchased under the NSO that are not vested at the time of the optionee’s cessationdirector’s termination of Boardcontinuous service.
The shares subject to each automatic NSO grant and each automatic RSU
award grant will immediately vest in full upon the director’s
termination of continuous service due to death or disability, or upon the occurrence of a change in control or hostile takeover
(as such terms are(each as defined in the 2018 Plan)
in each, subject to the director’s
continued Boardcontinuous service through
the date of such
date.Pursuant tochange in control or hostile takeover.
Accordingly, under the automatic grant program under the 2018 Plan, on the date of the
20182020 annual meeting of stockholders,
each of Mr. Carey, Dr. Dilly,
Dr. Jeffs, Dr. Mento,Mr. Meyers, Ms. Parker, Ms. Ramasastry,
Dr. Smith and Mr. Zakrzewski
each received an option to purchase
15,00020,000 shares of common stock with an exercise price per share of
$15.65,$10.46, and a RSU award of
2,50010,000 shares.
Under the terms of the automatic grant program under the 2018 Plan, On February 11, 2020, Dr. SmithMarkels was awarded an initial option to purchase 30,000 shares of our common stock on June 26, 2018, with an exercise price per share equal to the closing price per share of our common stock on that date. The option will vest in 36 successive equal monthly installments over the36-month period measured from the grant date, subject to Dr. Smith’s continued service as a member of the Board on each monthly vesting date. Also on June 26, 2018, Dr. Smith received$7.60 and a RSU award of 5,000 shares of common stock, pursuantand on September 9, 2020, Dr. Hillan was awarded an initial option to the automatic grant program under the 2018 Plan, which will vestpurchase 30,000 shares of our common stock with respect to 1/3rdan exercise price per share of the shares in three equal annual installments over the three-year period measured from the grant date, subject to Dr. Smith’s continued service as$10.43 and a member of the Board on each annual vesting date. The remaining terms of her options and RSU award are similar to the terms of the annual grants under the automatic grant program.
17
15,000 shares of common stock.
ADVISORY VOTE ON EXECUTIVE COMPENSATION
Under Section 14A(a)(1) of the Exchange Act, our stockholders are entitled to vote to approve, on a
non-binding advisory basis, the compensation of our named executive officers as disclosed in this Proxy Statement in accordance with SEC rules (commonly referred to as a
“say-on-pay” “say-on-pay” vote).
This vote is not intended to address any specific item of compensation, but rather the overall compensation of our named executive officers and the philosophy, policies and practices described in this Proxy Statement. Based on the voting results from the 2017 annual meeting of stockholders, our stockholders voted “For,” and we have adopted, an annual advisory vote on the compensation of the named executive officers in our proxy materials, which will remain in effect until the next required vote on the frequency of stockholder votes on the compensation of named executive officers.
Accordingly, the next advisory vote on the compensation of our named executive officers will be held at the 2022 Annual Meeting of Stockholders. Although the vote is
non-binding, the Board of Directors and the Compensation Committee value the opinions of our stockholders and will consider the outcome of the vote when making future compensation decisions affecting our executive officers.
We encourage stockholders to read closely the “Executive Compensation” section of this Proxy Statement, beginning with the Compensation Discussion and Analysis, or CD&A,
on page 28,included in the section labeled “Executive Compensation—Compensation Discussion and Analysis,” which describes in detail our executive compensation programs, policies and practices with respect to the compensation of our named executive officers in the year ended December 31,
2018.Our overarching compensation goal is to create a positive relationship between our compensation program and our corporate performance.
We design2020.
Resolution
As described in the CD&A, we believe that our executive compensation
program to implement our core objectives of attracting and retaining highly qualified executives with extensive industry or scientific experience, rewarding executives for success in meeting our annual clinical development and other research and operational goals, aligningprograms effectively align the
interestsinterest of our
executivesexecutive officers with those of our stockholders
and recognizing executives’ individualby linking a significant portion of their compensation to our corporate performance and
forby providing a competitive level of compensation designed to attract and retain highly qualified executives
other thancritical to our
Chief Executive Officer, achieving their individual goals and those ofsuccess. Accordingly, the
functional organizations that they manage.The Compensation Committee established the 2018 compensation program and the associated Company goals in accordance with the foregoing goals and philosophy. During 2018, we continued to maximize the application and value of our four technology platforms, as we work towards becoming a sustainable, fully integrated biopharmaceutical company. Our goal is to develop, manufacture and commercializes novel, proprietary genomic medicines that leverage our technical expertise, and strategically collaborate with industry partners when appropriate. In 2018, we formed new collaborations in central nervous system diseases and oncology with leading biopharmaceutical companies, acquired complementary technologies to enhance ourex vivo cell therapy capabilities in immunology and autoimmune diseases, made strong progress enrolling five active clinical trials, advanced additional preclinical assets toward an investigational new drug, or IND, application, and presented early data from the firstin vivo genome editing clinical trials in humans. The highlights of our business successes in 2018 are summarized under the heading “2018 and Early 2019 Performance Highlights” in the CD&A. The Board of Directors believes our executive compensation program uses appropriate structures and sound pay practices that are effective in achieving our core objectives.
Resolution
The Board is asking our stockholders to indicate their support for the compensation of our named executive officers, as described in this Proxy Statement, by casting anon-binding advisory vote “FOR” the following resolution:
“Resolved that the Company’s stockholders hereby approve, on an advisory basis, the compensation of the Company’s named executive officers as disclosed in this Proxy Statement pursuant to the
18
compensation disclosure rules of the SEC, including the Compensation Discussion and Analysis, the various compensation tables and the accompanying narrative discussion and any related material included in this Proxy Statement.”
Advisory approval of this Proposal No. 2 requires votes “FOR” from holders of a majority in voting power of the shares present
in persononline or represented by proxy at the Annual Meeting and entitled to vote on this
subject matter.Recommendation of the Board of Directors
Proposal No. 2.
RECOMMENDATION OF THE BOARD OF DIRECTORS
The Board of Directors recommends an advisory vote FOR the resolution to approve, on an advisory basis, the compensation of the named executive officers as disclosed in this Proxy Statement pursuant to the compensation disclosure rules of the SEC.
19
APPROVAL OF THE 2020 EMPLOYEE STOCK PURCHASE PLAN
We are asking our stockholders to approve the Sangamo Therapeutics, Inc. 2020 Employee Stock Purchase Plan, or the 2020 ESPP, at the Annual Meeting. The 2020 ESPP was approved by our Compensation Committee on September 18, 2020, subject to approval by our stockholders.
Approval of the 2020 ESPP will allow us to provide our employees with the opportunity to acquire an ownership interest in the Company through their participation in the 2020 ESPP, thereby encouraging them to remain in our service and more closely aligning their interests with those of our stockholders.
If this Proposal No. 3 is approved by our stockholders, the maximum number of shares of our common stock that may be issued under the 2020 ESPP will be 5,000,000 shares, subject to adjustment for certain changes in our capitalization. We do not maintain any other employee stock purchase plans, except the Sangamo Therapeutics, Inc. 2010 Employee Stock Purchase Plan, or the 2010 ESPP. The 2010 ESPP expired on April 30, 2020, but as of March 31, 2021, a total of 2,483,218 shares of our common stock were available for issuance pursuant to outstanding offerings under the 2010 ESPP. As of March 31, 2021, a total of 143,713,525 shares of our common stock were outstanding.
The 2020 ESPP became effective upon the date it was adopted by our Compensation Committee, but no purchase rights may be exercised under the 2020 ESPP unless this Proposal No. 3 is approved by our stockholders.
Description of the 2020 Employee Stock Purchase Plan
The material features of the 2020 ESPP are described below. The following description of the 2020 ESPP is a summary only and is qualified in its entirety by reference to the complete text of the 2020 ESPP. Stockholders are urged to read the actual text of the 2020 ESPP in its entirety, which is attached to this Proxy Statement as Appendix A.
Purpose
The purpose of the 2020 ESPP is to provide a means by which our employees may be given an opportunity to purchase shares of our common stock, to assist us in retaining the services of our employees, to secure and retain the services of new employees and to provide incentives for such persons to exert maximum efforts for our success.
The 2020 ESPP permits us to grant purchase rights that are intended to qualify as options issued under an “employee stock purchase plan” (as defined in Section 423(b) of the Internal Revenue Code of 1986, as amended, or the Code) (under the “423 Component” of the 2020 ESPP), as well as purchase rights that are not intended to so qualify (under the “Non-423 Component” of the 2020 ESPP).
Administration
Our Compensation Committee will administer the 2020 ESPP, unless otherwise determined by our Board. Our Board also retains concurrent authority to administer the 2020 ESPP. Our Compensation Committee will also have the power to delegate to a subcommittee any of the administrative powers our Compensation Committee is authorized to exercise under the 2020 ESPP, and in such case, our Compensation Committee retains the authority to concurrently administer the 2020 ESPP with any such subcommittee. Our Compensation Committee, our Board and any such subcommittee are each considered to be a Plan Administrator for purposes of this Proposal No. 3. The Plan Administrator has the final power to construe and interpret both the 2020 ESPP and the rights granted under it. The Plan Administrator has the power, subject to the provisions of the 2020 ESPP, to determine how and when rights to purchase our common stock will be granted, the provisions of each offering of such rights (which need not be identical), and whether employees of any of our parent or subsidiary companies will be eligible to participate in the 2020 ESPP.
Stock Subject to 2020 ESPP
Subject to adjustment for certain changes in our capitalization, the maximum number of shares of our common stock that may be issued under the 2020 ESPP will be 5,000,000 shares. If any rights granted under the 2020 ESPP terminate without being exercised in full, the shares of common stock not purchased under such rights will again become available for issuance under the 2020 ESPP. The shares of common stock issuable under the 2020 ESPP will be shares of authorized but unissued or reacquired common stock, including shares repurchased by us on the open market.
Offerings
The 2020 ESPP will be implemented by offerings of rights to purchase our common stock to all eligible employees. The Plan Administrator will determine the duration of each offering period, provided that in no event may an offering period exceed 27 months. The Plan Administrator may establish separate offerings which vary in terms (although not inconsistent with the provisions of the 2020 ESPP or the requirements of applicable laws). Each offering period will have one or more purchase dates, as determined by the Plan Administrator prior to the commencement of the offering period. The Plan Administrator has
the authority to alter the terms of an offering prior to the commencement of the offering period, including the duration of subsequent offering periods. When an eligible employee elects to join an offering period, he or she is granted a right to purchase shares of our common stock on each purchase date within the offering period. On the purchase date, all contributions collected from the participant are automatically applied to the purchase of our common stock, subject to certain limitations (which are described further below under “Eligibility”).
The Plan Administrator has the discretion to structure an offering so that if the fair market value of our common stock on the first trading day of a new purchase period within the offering period is less than or equal to the fair market value of our common stock on the first day of the offering period, then that offering will terminate immediately as of that first trading day, and the participants in such terminated offering will be automatically enrolled in a new offering beginning on the first trading day of such new purchase period.
Eligibility
Any individual who is employed by us (or by any of our parent or subsidiary companies if such company is designated by the Plan Administrator as eligible to participate in the 2020 ESPP) may participate in offerings under the 2020 ESPP, provided that such individual has been employed by us (or our parent or subsidiary, if applicable) for such continuous period preceding the first day of the offering period as the Plan Administrator may require (except as the Plan Administrator may provide with respect to newly eligible employees or as required by applicable law), but in no event may the required period of continuous employment be equal to or greater than two years. In addition, the Plan Administrator (unless prohibited by law) may provide that an employee will not be eligible to be granted purchase rights under the 2020 ESPP unless such employee is customarily employed for more than 20 hours per week and five months per calendar year. The Plan Administrator (unless prohibited by law) may also provide in any offering that certain of our employees who are “highly compensated” as defined in the Code are not eligible to participate in the 2020 ESPP. Furthermore, in the case of any offering under the Non-423 Component of the 2020 ESPP, an eligible employee may be excluded from participation in the 2020 ESPP if the Plan Administrator has determined that the participation of such eligible employee is not advisable or practical for any reason.
No employee will be eligible to participate in the 2020 ESPP if, immediately after the grant of purchase rights, the employee would own, directly or indirectly, stock possessing 5% or more of the total combined voting power or value of all classes of our stock or of any of our parent or subsidiary companies, including any stock which such employee may purchase under all outstanding purchase rights and options. In addition, no employee may purchase more than $25,000 worth of our common stock (determined based on the fair market value of the shares at the time such rights are granted) under all our employee stock purchase plans and any employee stock purchase plans of our parent or subsidiary companies for each calendar year during which such rights are outstanding.
As of March 31, 2021, we had approximately 430 employees.
Participation in the 2020 ESPP
An eligible employee may enroll in the 2020 ESPP by delivering to us, prior to the date selected by the Plan Administrator as the beginning of an offering period, an agreement authorizing contributions which may not exceed the maximum amount specified by the Plan Administrator. Each participant will be granted a separate purchase right for each offering in which he or she participates. Unless an employee’s participation is discontinued, his or her purchase right will be exercised automatically at the end of each purchase period at the applicable purchase price.
Purchase Price
The purchase price per share at which shares of our common stock are sold on each purchase date during an offering period will not be less than the lower of (i) 85% of the fair market value of a share of our common stock on the first day of the offering period or (ii) 85% of the fair market value of a share of our common stock on the purchase date.
As of March 31, 2021, the closing price of our common stock as reported on the Nasdaq Global Select Market was $12.53 per share.
Payment of Purchase Price; Contributions
The purchase of shares during an offering period generally will be funded by a participant’s payroll deductions accumulated during the offering period. If specifically provided in an offering, in addition to or instead of making contributions by payroll deductions, a participant may make contributions by cash, check or such other payment method specified by the Plan Administrator. A participant may change his or her rate of contributions, as determined by the Plan Administrator in the offering. All contributions made for a participant are credited to his or her account under the 2020 ESPP and deposited with our general funds, except where applicable law requires that contributions be deposited with a third party. Unless otherwise specified in the offering or required by applicable law, we will have no obligation to pay interest on contributions.
Purchase Limits
In connection with each offering made under the 2020 ESPP, the Plan Administrator may specify (i) a maximum number of shares of our common stock that may be purchased by any participant on any purchase date during such offering, (ii) a maximum aggregate number of shares of our common stock that may be purchased by all participants pursuant to such offering and/or (iii) a maximum aggregate number of shares of our common stock that may be purchased by all participants on any purchase date under such offering. If the aggregate number of shares of our common stock issuable upon exercise of purchase rights granted under the offering would exceed any such maximum aggregate number, then the Plan Administrator will make a pro rata allocation of available shares in a uniform and equitable manner.
Withdrawal
Participants may withdraw from a given offering by delivering a withdrawal form to us and terminating their contributions. Such withdrawal may be elected at any time prior to the end of an offering, except as otherwise provided by the Plan Administrator. Upon such withdrawal, such employee’s right to participate in that offering will terminate, and we will distribute as soon as practicable to the employee his or her accumulated but unused contributions. However, an employee’s withdrawal from an offering does not affect such employee’s eligibility to participate in subsequent offerings under the 2020 ESPP.
Termination of Eligibility
Unless otherwise required by applicable law, a participant’s rights under any offering under the 2020 ESPP will terminate immediately if the participant either (i) is no longer employed by us or any of our parent or subsidiary companies (subject to any post-employment participation period required by law) or (ii) is otherwise no longer eligible to participate. In such event, we will distribute as soon as practicable to such individual his or her accumulated but unused contributions.
Restrictions on Transfer
Rights granted under the 2020 ESPP are not transferable except by will, by the laws of descent and distribution, or if permitted by us, by a beneficiary designation. During a participant’s lifetime, such rights may only be exercised by the participant.
Changes in Capitalization
In the event of certain changes in our capitalization, the Plan Administrator will appropriately adjust: (i) the class(es) and maximum number of securities subject to the 2020 ESPP; (ii) the class(es) and number of securities subject to, and the purchase price applicable to outstanding purchase rights; and (iii) the class(es) and number of securities that are the subject of any individual and aggregate purchase limits under each ongoing offering.
Effect of Certain Corporate Transactions
In the event of a corporate transaction (as defined in the 2020 ESPP and described below), (i) any surviving or acquiring corporation (or its parent company) may assume or continue outstanding purchase rights granted under the 2020 ESPP or may substitute similar rights (including a right to acquire the same consideration paid to the stockholders in the corporate transaction) for such outstanding purchase rights, or (ii) if any surviving or acquiring corporation (or its parent company) does not assume or continue such purchase rights or does not substitute similar rights for such purchase rights, then the participants’ accumulated contributions will be used to purchase shares of our common stock within ten business days prior to the corporate transaction under such purchase rights, and such purchase rights will terminate immediately after such purchase.
For purposes of the 2020 ESPP, a corporate transaction generally will be deemed to occur in the event of the consummation of: (i) a sale or other disposition of all or substantially all of our consolidated assets; (ii) a sale or other disposition of more than 50% of our outstanding securities; (iii) a merger, consolidation or similar transaction following which we are not the surviving corporation; or (iv) a merger, consolidation or similar transaction following which we are the surviving corporation but the shares of our common stock outstanding immediately prior to such transaction are converted or exchanged into other property by virtue of such transaction.
Duration, Amendment and Termination
The Plan Administrator may amend or terminate the 2020 ESPP at any time. However, except in regard to certain capitalization adjustments, any such amendment must be approved by our stockholders if such approval is required by applicable law or listing requirements.
Any outstanding purchase rights granted before an amendment or termination of the 2020 ESPP will not be materially impaired by any such amendment or termination, except (i) with the consent of the employee to whom such purchase rights
were granted, (ii) as necessary to comply with applicable laws, listing requirements or governmental regulations (including Section 423 of the Code), or (iii) as necessary to obtain or maintain favorable tax, listing or regulatory treatment.
Notwithstanding anything in the 2020 ESPP or any offering to the contrary, the Plan Administrator will be entitled to: (i) establish the exchange ratio applicable to amounts withheld in a currency other than U.S. dollars; (ii) permit contributions in excess of the amount designated by a participant in order to adjust for mistakes in the processing of properly completed contribution elections; (iii) establish reasonable waiting and adjustment periods and/or accounting and crediting procedures to ensure that amounts applied toward the purchase of our common stock for each participant properly correspond with amounts withheld from the participant’s contributions; (iv) amend such purchase rights or clarify any ambiguities regarding the terms of any offering to enable such purchase rights to qualify under and/or comply with Section 423 of the Code with respect to the 423 Component of the 2020 ESPP; and (v) establish other limitations or procedures as the Plan Administrator determines in its sole discretion advisable that are consistent with the 2020 ESPP. Any such actions by the Plan Administrator will not be considered to alter or impair any purchase rights granted under an offering as they are part of the initial terms of each offering and the purchase rights granted under each offering.
U.S. Federal Income Tax Consequences
The following is a summary of the principal United States federal income tax consequences to participants and us with respect to participation in the 2020 ESPP. This summary is not intended to be exhaustive and does not discuss the income tax laws of any local, state or foreign jurisdiction in which a participant may reside. The information is based upon current federal income tax rules and therefore is subject to change when those rules change. Because the tax consequences to any participant may depend on his or her particular situation, each participant should consult the participant’s tax adviser regarding the federal, state, local, and other tax consequences of the grant or exercise of a purchase right or the sale or other disposition of common stock acquired under the 2020 ESPP. The 2020 ESPP is not qualified under the provisions of Section 401(a) of the Code and is not subject to any of the provisions of the Employee Retirement Income Security Act of 1974, as amended. Our ability to realize the benefit of any tax deductions described below depends on our generation of taxable income as well as the requirement of reasonableness and the satisfaction of our tax reporting obligations.
423 Component
Rights granted under the 423 Component of the 2020 ESPP are intended to qualify for favorable federal income tax treatment associated with rights granted under an employee stock purchase plan which qualifies under the provisions of Section 423 of the Code.
A participant will be taxed on amounts withheld for the purchase of shares of our common stock as if such amounts were actually received. Otherwise, under the 423 Component, no income will be taxable to a participant as a result of the granting or exercise of a purchase right until a sale or other disposition of the acquired shares. The taxation upon such sale or disposition will depend upon the holding period of the acquired shares.
Under the 423 Component, if the shares are sold or otherwise disposed of more than two years after the beginning of the offering period and more than one year after the shares are transferred to the participant, then the lesser of the following will be treated as ordinary income: (i) the excess of the fair market value of the shares at the time of such sale or disposition over the purchase price; or (ii) the excess of the fair market value of the shares as of the beginning of the offering period over the purchase price (determined as of the beginning of the offering period). Any further gain or any loss will be taxed as a long-term capital gain or loss.
Under the 423 Component, if the shares are sold or otherwise disposed of before the expiration of either of the holding periods described above, then the excess of the fair market value of the shares on the purchase date over the purchase price will be treated as ordinary income at the time of such sale or disposition. The balance of any gain will be treated as capital gain. Even if the shares are later sold or otherwise disposed of for less than its fair market value on the purchase date, the same amount of ordinary income is attributed to the participant, and a capital loss is recognized equal to the difference between the sales price and the fair market value of the shares on such purchase date. Any capital gain or loss will be short-term or long-term, depending on how long the shares have been held.
There are no federal income tax consequences to us by reason of the grant or exercise of rights under the 423 Component. We are entitled to a deduction to the extent amounts are taxed as ordinary income to a participant for shares sold or otherwise disposed of before the expiration of the holding periods described above (subject to the requirement of reasonableness and the satisfaction of tax reporting obligations).
Non-423 Component
Under the Non-423 Component, the excess of the fair market value of the shares on the purchase date over the purchase price will be treated as ordinary income at the time of such purchase. The amount of such ordinary income will be added to the participant’s basis in the shares, and any additional gain or resulting loss recognized on the disposition of the
shares after such basis adjustment will be a capital gain or loss. Any capital gain or loss will be short-term or long-term, depending on how long the shares have been held.
Under the Non-423 Component, we are entitled to a deduction in the year of purchase equal to the amount of ordinary income realized by a participant as a result of such purchase (subject to the requirement of reasonableness and the satisfaction of tax reporting obligations).
New Plan Benefits under 2020 ESPP
The following table sets forth certain information regarding benefits under the 2020 ESPP.
| | | | | | | | |
| Name and Position | | Number of
Shares |
Alexander D. Macrae President and Chief Executive Officer | | (1) |
Sung H. Lee Former Executive Vice President and Chief Financial Officer | | — (3) |
Gary H. Loeb Executive Vice President, General Counsel, Corporate Secretary and Chief Compliance Officer | | (1) |
D. Mark McClung Executive Vice President, Chief Business Officer | | (1) |
R. Andrew Ramelmeier Executive Vice President, Technical Operations | | (1) |
Stéphane Boissel Former Executive Vice President, Corporate Strategy | | — (2) |
| All current executive officers as a group | | (1) |
| All current directors who are not executive officers as a group | | — (4) |
| All current employees, including all current officers who are not executive officers, as a group | | (1) |
___________________(1)Participation in the 2020 ESPP is voluntary, and each eligible employee will make his or her own decision regarding whether and to what extent to participate in the 2020 ESPP. However, our Compensation Committee has granted purchase rights under the 2020 ESPP that are subject to stockholder approval of this Proposal No. 3. The number of shares of our common stock that may be purchased pursuant to such purchase rights will depend in part on the fair market value of our common stock on the applicable purchase date and, therefore, is not determinable at this time. However, as of March 31, 2021, there were 219 participants in the 2020 ESPP (including Mr. McClung and Dr. Ramelmeier) and, under the terms of the current offering, the maximum number of shares of our common stock that may be purchased by each participant on any purchase date is 5,000 shares. As of March 31, 2021, Dr. Macrae and Mr. Loeb do not hold any such purchase rights and have not elected to participate in the 2020 ESPP.
(2)Mr. Boissel resigned from his position as Executive Vice President, Corporate Strategy effective July 31, 2020 and, therefore, he was not granted any purchase rights under the 2020 ESPP and is not eligible to participate in the 2020 ESPP.
(3)Mr. Lee resigned from his position as Executive Vice President and Chief Financial Officer effective February 1, 2021 and, therefore, he does not hold any outstanding purchase rights under the 2020 ESPP and is not eligible to participate in the 2020 ESPP.
(4)Our non-employee directors are not eligible to participate in the 2020 ESPP.
Plan Benefits under 2020 ESPP
The following table sets forth certain information regarding purchase rights that have been granted under the 2020 ESPP as of March 31, 2021.
| | | | | |
| Name and Position | Number of
Shares |
Alexander D. Macrae President and Chief Executive Officer | — (1) |
Sung H. Lee Former Executive Vice President and Chief Financial Officer | — (3) |
Gary H. Loeb Executive Vice President, General Counsel, Corporate Secretary and Chief Compliance Officer | — (1) |
D. Mark McClung Executive Vice President, Chief Business Officer | (1) |
R. Andrew Ramelmeier Executive Vice President, Technical Operations | (1) |
Stéphane Boissel Former Executive Vice President, Corporate Strategy | — (2) |
| All current executive officers as a group | (1) |
| All current directors who are not executive officers as a group | — (4) |
| Each nominee for election as a director | |
| Alexander D. Macrae | — (1) |
| Robert F. Carey | — (4) |
| Kenneth Hillan | — (4) |
| John H. Markels | — (4) |
| James R. Meyers | — (4) |
| H. Stewart Parker | — (4) |
| Saira Ramasastry | — (4) |
| Karen L. Smith | — (4) |
| Joseph S. Zakrzewski | — (4) |
| Each associate of any executive officers, current directors or director nominees | — |
| Each other person who received or is to receive 5% of purchase rights | — |
| All current employees, including all current officers who are not executive officers, as a group | (1) |
___________________(1)As of March 31, 2021, no shares of our common stock have been purchased under the 2020 ESPP, and no shares of our common stock may be purchased under the 2020 ESPP unless this Proposal No. 3 is approved by our stockholders. Our Compensation Committee has granted purchase rights under the 2020 ESPP that are subject to stockholder approval of this Proposal No. 3. For more information regarding such purchase rights, please see footnote (1) to the table under “New Plan Benefits under 2020 ESPP” above. As of March 31, 2021, Dr. Macrae and Mr. Loeb do not hold any such purchase rights and have not elected to participate in the 2020 ESPP.
(2)Mr. Boissel resigned from his position as Executive Vice President, Corporate Strategy effective July 31, 2020 and, therefore, he was not granted any purchase rights under the 2020 ESPP and has not purchased any shares of our common stock under the 2020 ESPP.
(3)Mr. Lee resigned from his position as Executive Vice President and Chief Financial Officer effective February 1, 2021 and, therefore, he does not hold any outstanding purchase rights under the 2020 ESPP and has not purchased any shares of our common stock under the 2020 ESPP.
(4)Our non-employee directors are not eligible to participate in the 2020 ESPP.
Vote Required
Approval of this Proposal No. 3 requires votes “FOR” from holders of a majority in voting power of the shares present online or represented by proxy at the Annual Meeting and entitled to vote on Proposal No. 3.
RECOMMENDATION OF THE BOARD OF DIRECTORS
The Board of Directors recommends that the stockholders vote FOR the approval of the Sangamo Therapeutics, Inc. 2020 Employee Stock Purchase Plan.
PROPOSAL NO. 4:
RATIFICATION OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Audit Committee has appointed Ernst & Young LLP, our independent registered public accounting firm during
2018,2020, to serve in the same capacity for the year ending December 31,
2019,2021 and is asking the stockholders to ratify this appointment.
Neither our Bylaws nor other governing documents or law require stockholder ratification of the selection of Ernst & Young LLP as our independent registered public accounting firm. However, the Audit Committee is submitting the selection of Ernst & Young LLP to the stockholders for ratification as a matter of good corporate practice. In the event the stockholders fail to ratify the appointment, the Audit Committee will reconsider its selection. Even if the selection is ratified, the Audit Committee in its discretion may direct the appointment of a different independent registered public accounting firm at any time during the year if the Audit Committee believes that such a change would be in the best interests of Sangamo and its stockholders.
A representative of Ernst & Young LLP is expected to be present at the Annual Meeting and will have the opportunity to make a statement if he or she desires to do so and will be available to respond to appropriate questions.
Principal Accounting Fees and Services
The following table presents fees for professional audit services rendered by Ernst & Young LLP for the audit of our annual financial statements for 20182020 and 2017,2019:
| | | | | | | | | | | | | | |
| | Year Ended December 31, |
| | 2020 | | 2019 |
Audit fees and expenses(1) | | $ | 1,831,183 | | | $ | 1,520,256 | |
| Audit-related fees | | — | | | — | |
Tax fees(2) | | 74,720 | | | 26,780 | |
| All other fees | | — | | | — | |
| Total | | $ | 1,905,903 | | | $ | 1,547,036 | |
___________________
(1)Includes fees and expenses for the audit of our annual financial statements included in our annual reports on Form 10-K and the related audit of internal control over financial reporting, review of interim financial statements included in our quarterly reports on Form 10-Q, consultations regarding accounting and auditing matters, fees in connection with the filing of our registration statements on Form S-3 and Form S-8 and related amendments and services normally provided in connection with statutory and regulatory filings.
(2)Consists of fees billed for otherprofessional services rendered by Ernst & Young LLP during 2018for tax compliance, tax advice and 2017: | | | | | | | | | | | | |
| | | 2018 | | | 2017 | | | | |
Audit fees and expenses (1) | | $ | 2,205,461 | | | $ | 1,415,280 | | | | | |
Audit - related fees | | | - | | | | - | | | | | |
Tax fees (2) | | | 46,350 | | | | 43,350 | | | | | |
All other fees | | | - | | | | - | | | | | |
| | | | | | | | | | | | |
Total | | $ | 2,251,811 | | | $ | 1,458,630 | | | | | |
| | | | | | | | | | | | |
(1) | Includes fees and expenses for the audit of our annual financial statements included in our annual reports on Form10-K and the related audit of internal control over financial reporting, review of interim financial statements included in our quarterly reports on Form10-Q, consultations regarding accounting and auditing matters, fees in connection with our potential future public offering of shares of our common stock under the “at the market” agreement, fees in connection with the filing of our registration statements on FormS-3 and FormS-8 and related amendments and services normally provided in connection with statutory and regulatory filings.
|
(2) | Consists of fees billed for professional services for tax compliance, tax advice and tax planning.
|
tax planning.
Policy on Audit Committee
Pre-Approval of Audit and Permissible
Non-Audit Services
Under its charter, the Audit Committee must
pre-approve all engagements of the independent registered public accounting firm for the performance of all audit and
non-audit services that are not prohibited and the fees for such services. The Audit Committee has delegated to its
ChairmanChair the authority to evaluate and approve service engagements on behalf of the full committee in the event a need arises for specific
pre-approval between committee meetings. If the
ChairmanChair approves any such engagements, he will report such approval to the full Audit Committee not later than the next committee meeting.
20
The Audit Committee has determined that the rendering of other professional services for tax compliance and tax advice by Ernst & Young LLP is compatible with maintaining their independence. The Audit Committee has established a policy governing our use of Ernst & Young LLP fornon-audit services. Under the policy, management may use Ernst & Young LLP fornon-audit services that are permitted under SEC rules and regulations, provided that management obtains the Audit Committee’s approval before such services are rendered.
The services provided by Ernst & Young LLP in
20182020 were
pre-approved in accordance with this policy.
Approval of this Proposal No.
34 requires votes “FOR” from holders of a majority in voting power of the shares present
in persononline or represented by proxy at the Annual Meeting and entitled to vote on this
subject matter.Recommendation of the Board of Directors
Proposal No. 4.
RECOMMENDATION OF THE BOARD OF DIRECTORS
The Board of Directors recommends that the stockholders vote FOR the ratification of the appointment of Ernst & Young LLP to serve as our independent registered public accounting firm for the year ending December 31,
2019.21
2021.
OTHER MATTERS
The Board knows
The following table sets forth information regarding our
current executive officers as of
April 25, 2019:March 31, 2021: | | | | | | | | | | | | | | |
Name | | Age | | | Position |
Alexander D. Macrae, M.B., Ch.B., Ph.D. | | 58 | 56 | | | President, Chief Executive Officer and Director |
Kathy Y. Yi
Gary H. Loeb | | 51 | 48 | | | Executive Vice President, General Counsel, Corporate Secretary and Chief FinancialCompliance Officer (Principal Financial and Accounting Officer) |
Stéphane Boissel
D. Mark McClung | | 58 | 51 | | | Executive Vice President, Corporate StrategyChief Business Officer |
Adrian Woolfson,R. Andrew Ramelmeier, Ph.D.
| | 59 | 54 | | | Executive Vice President, ResearchTechnical Operations |
| Jason D. Fontenot, Ph.D. | | 51 | | | Senior Vice President, Chief Scientific Officer |
| Robert J. Schott, M.D., M.P.H., F.A.C.C. | | 63 | | | Senior Vice President, Head of Development |
| Prathyusha Duraibabu | | 42 | | | Vice President, Finance; Principal Financial Officer; and DevelopmentPrincipal Accounting Officer |
Alexander D. Macrae. Biographical information regarding Dr. Macrae is set forth under “Proposal No. 1: Election of Directors.”Kathy Y. Yi
Gary H. Loeb has served as our Executive Vice President and General Counsel since July 2019 and as our Corporate Secretary and Chief Compliance Officer since September 2019. Mr. Loeb is responsible for all legal matters for Sangamo and has over 20 years of experience in biotechnology, legal transactions and operations, and compliance. Prior to joining Sangamo, Mr. Loeb served from November 2016 to June 2019 as General Counsel, Corporate Secretary and Chief Compliance Officer at Achaogen Inc., a public biopharmaceutical company. As a member of the Executive Team, he built Achaogen’s legal and compliance functions and supported its first product launch for plazomicin, which is listed by the World Health Organization as an “essential medicine.” In April 2019, Achaogen filed a voluntary petition for relief under Chapter 11 of the U.S. Bankruptcy Code and most of its assets were subsequently sold to a multinational pharmaceutical company. Prior to joining Achaogen, Mr. Loeb served as General Counsel for Sano Intelligence Inc., a medical device company from November 2015 to June 2016, and as Executive Vice President, Business Development and General Counsel of Counsyl, Inc., a health technology company, from 2013 to 2015. Prior to Counsyl, Mr. Loeb was General Counsel at Amyris, Inc., a public synthetic biotechnology company and Vice President of Intellectual Property at Genentech, Inc, where he had roles of increasing responsibility over eleven years. Mr. Loeb received his B.S. and B.A. degrees from Stanford University and his J.D. from Columbia Law School.
D. Mark McClunghas served as our Executive Vice President and Chief FinancialBusiness Officer since December 2018,May 2020. Mr. McClung is responsible for corporate and business development, product strategy and planning, alliance management, patient advocacy, government relations, investor relations, corporate communications and facilities. From February 2019 until joining Sangamo, Mr. McClung consulted to the biopharmaceutical industry, including Sangamo. Prior thereto, from 2015 through February 2019, Mr. McClung was Vice President and General Manager of Global Oncology Commercial at Amgen Inc., a public biopharmaceutical company, which he joined our companyfollowing Amgen’s acquisition of Onyx Pharmaceuticals Inc., where he had served as Senior Vice President & Chief Commercial Officer. For two decades prior, Mr. McClung held roles of increasing responsibility at GlaxoSmithKline in marketing and Chief Financial Officersales, commercial operations, clinical development and product strategy, and general management in February 2017. Ms. Yi has over 18 years of experience in corporate finance,Canada, the United States, and Europe, including financial analysis in support of M&A transactions, licensingas Vice President and other business development activities. Prior to joining us, Ms. Yi served as Head of FinanceGlobal Commercial for Global Inhalation Technical Research & DevelopmentGSK Oncology from 2009 to 2013. Mr. McClung received his bachelor’s degree in Human Kinetics and Biomedical Sciences from the University of Guelph in Ontario, Canada and completed graduate coursework at Novartis Pharmaceutical Corporation since 2014. From 2007 to 2014, Ms. Yi held various financial management positions of increasing seniority at Life Technologies Corporation, a Nasdaq-listed life science company that was acquired by Thermo Fisher ScientificYork University in 2014, including Finance Leader, Corporate FP&A from 2012 to 2014, Director of Finance, M&A/Corporate Development from 2010 to 2012,Toronto and Director of Finance, Global Manufacturing Operations from 2007 to 2010. From 2001 to 2007, Ms. Yi held increasing roles of responsibilities in Corporate Finance at Intel Corporation. Ms. Yi began her career as a process engineer for Bechtel Corporation. Ms. Yi received a B.S. in Chemical EngineeringWharton Business School at the University of California at Berkeley and an M.B.A. from Columbia University.Stéphane BoisselPennsylvania.
R. Andrew “Andy” Ramelmeier, Ph.D.has served as our Executive Vice President, Technical Operations since September 2019 and is responsible for technical operations at Sangamo, including manufacturing, quality, supply chain and process and analytical development. Dr. Ramelmeier joined Sangamo in January 2018 as Senior Vice President, Technical Operations. Dr. Ramelmeier has 30 years of Corporate Strategy since October 2018. Prior thereto, since April 2015, Mr. Boisselexperience in the biopharmaceutical industry, developing and transferring biological processes, designing and building manufacturing facilities, and directing contract manufacturers as well as internal manufacturing operations. From 2014 through 2017, Dr. Ramelmeier served as Chief Executive OfficerSenior Vice President, Technical Operations at Portola Pharmaceuticals, Inc., a biopharmaceutical company, where he was responsible for tech transfer, bulk and drug product manufacturing, technical support and supply chain of TxCell S.A. which we acquired in October 2018. PriorPortola’s pipeline products. From 2006 to TxCell S.A., Mr. Boissel2014, Dr. Ramelmeier served as Chief Executive Officer of Genclis, a molecular diagnostics company from 2014 to April 2015. From 2010 to 2014, Mr. Boisel served as Executive Vice President, Manufacturing, Process Sciences and Chief Financial Officer of Trangene S.A.Facilities at BioMarin Pharmaceutical Inc., overseeing multiple commercial biologics products, clinical pipeline, and from 2002 to 2010, he served as Chief Financial Officerfacilities in Novato, California, and then Executive Vice President and Chief Financial Officer of Innate Pharma S.A.Shanbally, Ireland. Earlier in his career, Mr. Boissel worked in investment banking for Lazard, where he focused on principal investment in France, Singapore and Hong Kong. Mr. Boissel completed his undergraduate work in management and finance at the University of Lyon and Paris-Dauphine in France and received his M.B.A. from the University of Chicago.Adrian Woolfson, B.M., B.Ch., Ph.D.has served as our Executive Vice President of Research and Development since January 2019. Dr. Woolfson has over a decade of biopharmaceutical industry experience in drug discovery, medical affairs and early and late stage clinical development. Most recently, from January 2018 to November 2018, he served as Chief Medical Officer at Nouscom AG, a genetic cancer vaccine biotechnology company based in Basel, Switzerland, where he led the development of the company’soff-the-shelf and personalized neoantigen vaccine and oncolytic virus strategy. Prior to Nouscom, Dr. Woolfson served as Global Clinical Leader, Early and Late Stage Immuno-Oncology/Hematology at Pfizer, Inc. in New York from January 2015 to January 2018, and was responsible for defining Pfizer’s hematology immune-oncology strategy and building its immune-oncology hematological malignancies portfolio. Prior to that, he was the Global Lead from July 2013 to January 2018 for Pfizer’s smoothened, or SMO, inhibitor glasdegib, which received FDA approval in 2018. From 2007 to 2013, Dr. Woolfson held roles of increasing responsibility at Bristol-Myers Squibb,Johnson & Johnson and Merck & Co., Inc. Prior to joining industry, Dr. Ramelmeier conducted post-doctoral work in Germany. He received his B.S. in Chemical Engineering from Johns Hopkins and his Ph.D. in Chemical Engineering from the University of California, Berkeley.
Jason D. Fontenot, Ph.D. has served as our Senior Vice President, Chief Scientific Officer since January 2021. Dr. Fontenot oversees all platform and pipeline research activities – advancing Sangamo’s proprietary genomic medicine
technology in support of a diverse clinical pipeline. Dr. Fontenot joined Sangamo in March 2019 as Senior Vice President, Cell Therapy. In that role, Dr. Fontenot oversaw Sangamo’s engineered cell therapy portfolio, including
Global Medical Leada proprietary chimeric antigen receptor regulatory T cells pipeline, and partnerships with Sanofi S.A., or Sanofi, and Kite Pharma Inc., or Kite, a Gilead Company. Dr. Fontenot has an extensive background in cell therapy and drug development, having previously served as Chief Scientific Officer at Immusoft Corporation, a biotechnology company, from July 2018 to March 2019, Head of Exploratory Research at Juno Therapeutics Inc., a biopharmaceutical company, from January 2016 to January 2018 when it was acquired by Celgene Corporation, and for
nearly a
first-in-human CDC7 inhibitor and selective JAK2 inhibitor. Dr. Woolfson completed his post-graduate training in internal medicine at Addenbrooke’s Hospital, Cambridge decade as a group leader in the
United Kingdom.immunology department at Biogen Inc. Dr. Fontenot has expertise in lymphocyte biology, cell and genetic engineering and immune-mediated disease. Dr. Fontenot conducted post-doctoral work at Rockefeller University in New York City. He
holds a B.M. B.Ch. degree in Clinical Medicine from Oxford University and completedreceived his
23
Ph.D. in molecular immunology at Cambridge University. He wasImmunology from the Charles and Katherine Darwin Research Fellow at Darwin College, Cambridge and a Wellcome Trust Clinical Research Fellow. He completed his doctoral and post-doctoral workUniversity of Washington in Seattle working in the laboratory of Nobel Prize winnerDr. Alexander Rudensky.
Robert J. Schott, M.D., M.P.H., F.A.C.C., has served as our Senior Vice President, Head of Development since February 2021. Dr. Schott is responsible for leading our clinical development organization across all phases of drug development and inventorregulatory approval. Prior to joining Sangamo, Dr. Schott served from March 2017 to January 2021 as the Chief Medical Officer at Chorus, an autonomous early-phase development engine within Eli Lilly and Company, a public pharmaceutical company, where he oversaw teams working to expand Eli Lilly’s pipeline. He previously joined Eli Lilly in 2014 as a Global Launch Leader. Earlier in his career, Dr. Schott served as a Cardiovascular Department Chair and Vice President of monoclonal antibodies,Medical Affairs at Sutter Medical Center Sacramento while continuing to care for patients as an invasive cardiologist. Dr. Cesar MilsteinSchott received his B.S. in Biomedicine, M.D. and M.P.H. in Epidemiology from the University of Michigan. He trained in cardiovascular medicine at the Massachusetts General Hospital after working as a post-doctoral fellow at the Harvard Medical School.
Prathyusha Duraibabu has served as our Principal Financial Officer since March 2021, our Principal Accounting Officer since June 2019 and our Vice President, Finance since March 2019. Ms. Duraibabu has over 22 years of experience in optimizing financial operations, driving organizational change, building diverse teams and delivering results. Prior to joining us, Ms. Duraibabu served as Corporate Controller at Pacific Biosciences of California, Inc., a public commercial biotechnology company, from June 2010 to March 2019, where she was responsible for global financial operations, strategy, audit and tax. Ms. Duraibabu received her B.S. in Accounting from Oxford Brookes University in Oxford, United Kingdom, and her M.B.A. from San Jose State University. Ms. Duraibabu is a Certified Public Accountant in the DivisionState of Protein and Nucleic Acid Chemistry at the MRC Laboratory of Molecular Biology in Cambridge, United Kingdom.24
California.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information known to Sangamo with respect to the beneficial ownership of our common stock as of April 18, 2019,March 31, 2021, except as noted, by (i) all persons who were beneficial owners of five percent (5%)5% or more of our common stock based on 115,158,847143,713,525 shares outstanding as of April 18, 2019,March 31, 2021, (ii) each current director and(which includes all nominees for director), (iii) each nominee for director, (iii) theof our named executive officers, named in the Summary Compensation Table of the Executive Compensation of this Proxy Statement, and (iv) all current directors and executive officers as a group. Unless otherwise indicated, the principal address of each of the stockholders below is c/o Sangamo Therapeutics, Inc., 501 Canal7000 Marina Boulevard, Richmond,Brisbane, CA 94804.94005. Except as otherwise indicated or to the extent authority may be shared by both
spouses under applicable law, and subject to applicable community property laws, we believe the persons named in the table have sole voting and investment power with respect to all shares of common stock held by them.
| | | | | | | | |
Name and Address of Beneficial Owner | | Number of
Shares
Beneficially
Owned | | | Percentage
of Shares
Beneficially
Owned | |
BlackRock, Inc. (1) | | | | | | | | |
55 East 52nd Street New York, NY 10055 | | | 8,430,838 | | | | 7.3 | % |
Vanguard Group Inc. (2) 100 Vanguard Blvd. Malvern, PA 19355 | | | 7,595,252 | | | | 6.6 | % |
State Street Corporation (3) State Street Financial Center One Lincoln Street Boston, MA 02111 | | | 5,910,152 | | | | 5.1 | % |
Alexander D. Macrae (4) | | | 804,526 | | | | | * |
Stéphane Boissel | | | - | | | | | - |
Robert F. Carey (5) | | | 57,500 | | | | | * |
Edward R. Conner, M.D. (6) | | | 64,458 | | | | | * |
Stephen G. Dilly, M.B.B.S., Ph.D. (7) | | | 137,500 | | | | | * |
Roger Jeffs (8) | | | 54,908 | | | | | * |
Steven J. Mento, Ph.D. (9) | | | 101,546 | | | | | * |
H. Stewart Parker (10) | | | 102,500 | | | | | * |
Saira Ramasastry (11) | | | 67,500 | | | | | * |
Karen L. Smith (12) | | | 30,000 | | | | | * |
Heather D. Turner (13) | | | 54,166 | | | | | * |
Kathy Y. Yi (14) | | | 63,429 | | | | | * |
Joseph S. Zakrzewski (15) | | | 47,500 | | | | | * |
All current directors and executive officers as a group (12 persons) (16) | | | 1,490,589 | | | | 1.3 | % |
(1) | This information is based solely on information contained in the Schedule 13G/A filed with the SEC on February 6, 2019 by BlackRock, Inc., or BlackRock. BlackRock, as a parent holding company or control person, may be deemed to beneficially own the indicated shares and has sole dispositive power over 8,430,838 shares and sole voting power over 8,139,917 shares. BlackRock reported its beneficial ownership on behalf of itself and the following: BlackRock International Limited, BlackRock Advisors, LLC, BlackRock (Netherlands) B.V., BlackRock Institutional Trust Company, National Association, BlackRock Asset Management Ireland Limited, BlackRock Financial Management, Inc., BlackRock Japan Co., Ltd., BlackRock Asset Management Schweiz AG, BlackRock Investment Management, LLC, BlackRock Investment Management (UK) Limited, BlackRock Asset Management Canada Limited, BlackRock
|
25
| | | | | | | | | | | | | | | | | | | | |
| Name and Address of Beneficial Owner | | Number of
Shares
Beneficially
Owned | | Percentage
of Shares
Beneficially
Owned | | |
Biogen, Inc. (1) 225 Binney Street Cambridge, MA 02142 | | 24,420,157 | | | 17.0 | | | % |
Wasatch Advisors, Inc. (2) 505 Wakara Way Salt Lake City, UT 84108 | | 10,072,381 | | | 7.0 | | | % |
BlackRock, Inc. (3) 55 East 52nd Street New York, NY 10055 | | 9,566,415 | | | 6.7 | | | % |
Vanguard Group Inc. (4) 100 Vanguard Blvd. Malvern, PA 19355 | | 8,453,658 | | | 5.9 | | | % |
| Alexander D. Macrae (5) | | 1,611,535 | | | 1.1 | | | % |
| Stéphane Boissel (6) | | 3,449 | | | | | * |
| Robert F. Carey (7) | | 121,400 | | | | | * |
| Kenneth J. Hillan (8) | | 46,400 | | | | | * |
| Sung H. Lee (9) | | 9,540 | | | | | * |
| Gary H. Loeb (10) | | 134,547 | | | | | * |
| John H. Markels (11) | | 48,067 | | | | | * |
| D. Mark McClung | | — | | | | | * |
| James R. Meyers (12) | | 78,067 | | | | | * |
| H. Stewart Parker (13) | | 169,400 | | | | | * |
| Saira Ramasastry (14) | | 121,400 | | | | | |
| R. Andrew Ramelmeier (15) | | 199,721 | | | | | * |
| Karen L. Smith (16) | | 97,233 | | | | | * |
| Joseph S. Zakrzewski (17) | | 136,400 | | | | | * |
| All current directors and executive officers as a group (15 persons) (18) | | 2,957,631 | | | 2.1 | | | % |
___________________
*Less than 1%.
(1)This information is based solely on information contained in the Schedule 13G filed with the SEC on April 17, 2020 by Biogen Inc. and Biogen MA Inc. There have been no reported changes to the beneficial ownership of Biogen Inc. and Biogen MA, Inc. since April 8, 2020. We and Biogen MA, Inc., or BIMA, are party to a voting agreement in which BIMA has agreed that, excluding specified extraordinary matters, it will vote its shares in accordance with our recommendation and has granted us an irrevocable proxy with respect to the foregoing. Such voting provisions expire on the earlier of (i) April 9, 2022, (ii) the date that BIMA beneficially owns less than 5% of our common stock and (iii) the date our collaboration agreement with BIMA and Biogen International GmbH, which we refer to together as Biogen, is terminated; provided, however, that in no event shall such expiration date be prior to April 9, 2021. None of the proposals in this Proxy Statement are extraordinary matters for purposes of our voting agreement with BIMA and we intend to instruct BIMA to vote “For” each of the proposals, including for each nominee for director, pursuant to the voting agreement.
(2)This information is based solely on information contained in the Schedule 13G/A filed with the SEC on February 11, 2021 by Wasatch Advisors, Inc., or Wasatch. The Schedule 13G/A provides information only as of December 31, 2020 and, consequently, the beneficial ownership of Wasatch may have changed between December 31, 2020 and March 31, 2021.
(3)This information is based solely on information contained in the Schedule 13G/A filed with the SEC on February 1, 2021 by BlackRock, Inc., or BlackRock. BlackRock, as a parent holding company or control person, may be deemed to beneficially own the indicated shares and has sole dispositive power over 9,566,415 shares and sole voting power over 9,411,748 shares. BlackRock reported its beneficial ownership on behalf of itself and the following: BlackRock life Limited; BlackRock Advisors, LLC; BlackRock (Netherlands) B.V.; BlackRock Institutional Trust Company, National Association; BlackRock Asset Management Ireland Limited; BlackRock Financial Management, Inc.; BlackRock Japan Co., Ltd.; BlackRock Asset Management Schweiz AG; BlackRock Investment Management, LLC; BlackRock Investment Management (UK) Limited; BlackRock Asset Management Canada Limited; BlackRock Investment Management (Australia) Limited;, BlackRock Fund Advisors; and BlackRock Fund Managers Ltd. The Schedule 13G/A provides information only as of December 31, 2020 and, consequently, the beneficial ownership of the above-mentioned entities may have changed between December 31, 2020 and March 31, 2021.
| (Luxembourg) S.A., BlackRock Investment Management (Australia) Limited and BlackRock Fund Advisors. The Schedule 13G/A provides information only as of December 31, 2018 and, consequently, the beneficial ownership of the above-mentioned entities may have changed between December 31, 2018 and April 18, 2019.
|
(2) | This information is based solely on information contained in the Schedule 13G/A filed with the SEC on February 13, 2019 by The Vanguard Group, Inc., or Vanguard. Vanguard may be deemed to beneficially own the indicated shares and has sole dispositive power over 7,387,528 shares, shared dispositive power over 207,724 shares, sole voting power over 211,134 shares and shared voting power over 7,045 shares. Vanguard reported its beneficial ownership on behalf of itself and the following: Vanguard Fiduciary Trust Company and Vanguard Investments Australia, Ltd., each a wholly owned subsidiary of Vanguard. The Schedule 13G/A provides information only as of December 31, 2018 and, consequently, the beneficial ownership of the above-mentioned entities may have changed between December 31, 2018 and April 18, 2019.
|
(3) | This information is based solely on information contained in the Schedule 13G filed with the SEC on February 13, 2019 by State Street Corporation, or State Street. State Street may be deemed to beneficially own the indicated shares and has shared dispositive power over 5,910,152 shares and shared voting power over 5,572,841 shares. State Street reported its beneficial ownership on behalf of itself and the following: SSGA Funds Management, Inc., State Street Global Advisors Limited (UK), State Street Global Advisors, Australia Limited and State Street Global Advisors Trust Company. The Schedule 13G provides information only as of December 31, 2018 and, consequently, the beneficial ownership of the above-mentioned entities may have changed between December 31, 2018 and April 18, 2019.
|
(4) | Includes 798,000 shares of common stock subject to options exercisable within 60 days after April 18, 2019.
|
(5) | Consists of (i) 55,000 shares of common stock subject to options exercisable within 60 days after April 18, 2019 and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019.
|
(6) | Includes 57,250 shares of common stock subject to options exercisable within 60 days after April 18, 2019.
|
(7) | Consists of (i) 135,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019 and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019.
|
(8) | Includes (i) 45,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019, of which 34,166 shares fully vest within 60 days of April 18, 2019, and the remaining 10,834 shares are currently exercisable but do not vest within 60 days of April 18, 2019 and would be subject to repurchase upon cessation of service to the Board if exercised prior to vesting and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019. Dr. Jeffs is not standing for reelection to the Board of Directors at the Annual Meeting.
|
(9) | Includes (i) 55,000 shares of common stock subject to options exercisable within 60 days after April 18, 2019, (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019 and (iii) 32,214 shares held in a family trust, of which Dr. Mento isco-trustee.Table of Contents Dr. Mento is not standing for reelection to the Board of Directors at the Annual Meeting.
|
(10) | Includes (i) 95,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019 and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019.
|
(11) | Consists of (i) 65,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019 and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019.
|
(12) | Consists of 30,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019, of which 9,166 shares fully vest within 60 days of April 18, 2019, and the remaining 20,834
|
26
(4)This information is based solely on information contained in the Schedule 13G/A filed with the SEC on February 10, 2021 by The Vanguard Group, Inc., or Vanguard. Vanguard may be deemed to beneficially own the indicated shares and has sole dispositive power over 8,099,242 shares, shared dispositive power over 354,416 shares, and shared voting power over 262,029 shares. Vanguard reported its beneficial ownership on behalf of itself and the following: Vanguard Asset Management, Limited; Vanguard Fiduciary Trust Company; Vanguard Global Advisors, LLC; Vanguard Group (Ireland) Ltd; Vanguard Investments Australia, Ltd, Vanguar5d Investments Canada Inc.; Vanguard Investments Hong Kong Limited; and Vanguard investments UK, Limited. The Schedule 13G/A provides information only as of December 31, 2020 and, consequently, the beneficial ownership of the above-mentioned entities may have changed between December 31, 2020 and March 31, 2021.
(5)Includes 1,536,436 shares of common stock subject to options exercisable within 60 days after March 31, 2021.
(6)Mr. Boissel resigned from his position as Executive Vice President, Corporate Strategy effective July 31, 2020.
(7)Includes (i) 106,400 shares of common stock subject to options exercisable within 60 days after March 31, 2021, of which 94,100 shares fully vest within 60 days of March 31, 2021, and the remaining 12,300 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000 shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(8)Includes 46,400 shares of common stock subject to options exercisable within 60 days after March 31, 2021, of which 10,766 shares fully vest within 60 days of March 31, 2021, and the remaining 35,634 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting.
(9)Mr. Lee resigned from his position as Executive Vice President and Chief Financial Officer effective February 1, 2021.
(10)Includes 128,644 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021.
(11)Includes 46,400 shares of common stock subject to options exercisable within 60 days of March 31, 2021, of which 16,600 shares fully vest within 60 days of March 31, 2021, and the remaining 29,800 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting.
(12)Includes (i) 66,400 shares of common stock subject to options exercisable within 60 days of March 31, 2021 of which 39,100 shares fully vest within 60 days of March 31, 2021, and the remaining 27,300 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000 shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(13)Includes (i) 146,400 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021, of which 134,100 shares fully vest within 60 days of March 31, 2021, and the remaining 12,300 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000 shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(14)Includes (i) 106,400 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021, of which 94,100 shares fully vest within 60 days of March 31, 2021, and the remaining 12,300 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(15)Includes 176,562 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021.
(16)Consists of (i) 81,400 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021, of which 68,266 shares fully vest within 60 days of March 31, 2021, and the remaining 13,134 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000 shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(17)Includes (i) 96,400 shares of common stock issuable upon the exercise of stock options within 60 days of March 31, 2021, of which 84,100 shares fully vest within 60 days of March 31, 2021, and the remaining 12,300 shares are currently exercisable but do not vest within 60 days of March 31, 2021 and would be subject to repurchase upon cessation of continuous service if exercised prior to vesting and (ii) RSUs covering 10,000 shares of common stock that vest and become issuable within 60 days of March 31, 2021.
(18)The percentages are calculated based on 143,713,525 shares outstanding as of March 31, 2021. Shares of common stock issuable upon the exercise of options currently exercisable or exercisable within 60 days after March 31, 2021, are deemed outstanding for purposes of computing the percentage ownership of the person holding such options, but are not deemed outstanding for computing the percentage ownership of any other person. Because Mr. Boissel resigned effective July 31, 2020 and Mr. Lee resigned effective as of February 1, 2021, the number of shares and percentage ownership indicated in the table above with respect to the beneficial ownership of all directors and executive officers as a group do not include any shares beneficially owned by Mr. Boissel or Mr. Lee.
DELINQUENT SECTION 16(a) REPORTS
The members of the Board of Directors, our executive officers and persons who beneficially own more than 10% of our outstanding common stock are subject to the reporting requirements of Section 16 of the Exchange Act, as amended, which require them to file reports with respect to their beneficial ownership of the common stock and their transactions in such common stock. Based upon (i) a review of Forms 3 and 4 and any amendments thereto filed electronically with the SEC during 2020, (ii) a review of Forms 5 and any amendments thereto filed electronically with the SEC with respect to 2020, and (iii) written representations that no Form 5 reports were required, we believe that all reporting requirements under Section 16 for such year were met in a timely manner by our directors, executive officers and greater than 10% beneficial owners, except for the following: Dr. Ramelmeier filed two Form 4 reports late, each disclosing one transaction occurring in 2020; each of Mr. Carey, Dr. Dilly, Mr. Meyers, Ms. Parker, Ms. Ramasastry, Dr. Smith and Mr. Zakrzewski, filed an amendment to a
| shares are currently exercisable but do not vest within 60 days of April 18, 2019 and would be subject to repurchase upon cessation of service to the Board if exercised prior to vesting.
|
(13) | Consists of 54,166 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019 Ms. Turner resigned from her position as Executive Vice President and General Counsel effective as of March 15, 2019.
|
(14) | Includes 62,000 shares of common stock subject to options exercisable within 60 days after April 18, 2019.
|
(15) | Consists of (i) 45,000 shares of common stock issuable upon the exercise of stock options within 60 days of April 18, 2019, of which 34,166 shares fully vest within 60 days of April 18, 2019, and the remaining 10,834 shares are currently exercisable but do not vest within 60 days of April 18, 2019 and would be subject to repurchase upon cessation of service to the Board if exercised prior to vesting and (ii) RSUs covering 2,500 shares of common stock that vest and become issuable within 60 days of April 18, 2019.
|
(16) | The percentages are calculated based on 115,158,847 shares outstanding as of April 18, 2018. Shares of common stock issuable upon the exercise of options currently exercisable or exercisable within 60 days after April 18, 2019, are deemed outstanding for purposes of computing the percentage ownership of the person holding such options, but are not deemed outstanding for computing the percentage ownership of any other person. Because Ms. Turner resigned effective as of March 15, 2019 and Dr. Conner ceased being a Section 16 officer effective as of January 21, 2019, the number of shares and percentage ownership indicated in the table above with respect to the beneficial ownership of all directors and executive officers as a group do not include any shares beneficially owned by Ms. Turner or Dr. Conner. As Dr. Woolfson became a Section 16 officer effective as of January 21, 2019, shares beneficially owned by Dr. Woolfson are included in the table above with respect to the beneficial ownership of all directors and executive officers as a group.
|
27
previously filed Form 4 revising disclosures for two transactions occurring in 2020; and Biogen Inc. and Biogen MA Inc. filed one Form 3 late disclosing one transaction occurring in 2020.
EXECUTIVE COMPENSATION
COMPENSATION DISCUSSION AND ANALYSIS
Compensation Discussion and Analysis
It is our intent in this CD&A to inform our stockholders of the policies and objectives underlying the compensation programs for our executive officers. Accordingly, we will address and analyze the key elements of the compensation provided to our principal executive officer,
our principal financial officer
andduring 2020, our three other most highly compensated executive officers as of December 31,
2018.2020 and a former highly compensated executive officer who was not serving as an executive officer as of December 31, 2020. We refer to these officers as our
named“named executive officers.
” | | | | | | | | |
Alexander D. Macrae | | President and Chief Executive Officer |
Kathy Y. Yi
Sung H. Lee (1) | | Former Executive Vice President and Chief Financial Officer |
Heather D. Turner
Gary H. Loeb | | Former Executive Vice President, General Counsel, Corporate Secretary and SecretaryChief Compliance Officer |
Stéphane Boissel
D. Mark McClung (2) | | Executive Vice President, Chief Business Officer |
| R. Andrew Ramelmeier | | Executive Vice President, Corporate DevelopmentTechnical Operations |
Edward R. Conner, M.D.
Stéphane Boissel (3) | | SeniorFormer Executive Vice President, and Chief Medical OfficerCorporate Strategy |
___________________
(1)Mr. Lee resigned effective February 1, 2021.
(2)Mr. McClung commenced employment with us effective May 1, 2020.
(3)Mr. Boissel resigned effective July 31, 2020.
We are a clinical stage biotechnologygenomic medicine company focused oncommitted to translating ground-breaking science into genomic medicines with the potential to transform patients’ lives using our platform technologies in genome editing, gene therapy, cell therapy and genome engineering. We plan to deliver on this mission through (i) development of our clinical and preclinical product candidates, (ii) our novel science and (iii) our in-house manufacturing capabilities.
Our Product Candidates
Our current clinical-stage product candidates are:
•Giroctocogene fitelparvovec, also known as SB-525, our lead product candidate, is a gene regulationtherapy for the treatment of hemophilia A and cell therapy.is currently being evaluated in the registrational Phase 3 AFFINE (efficAcy and saFety Factor vIii geNe thErapy) clinical trial. We are focused on threedeveloping giroctocogene fitelparvovec with our collaborator Pfizer Inc., or Pfizer;
•ST-920, our wholly-owned gene therapy product candidate for the treatment of Fabry disease, is currently being evaluated in our Phase 1/2 STAAR clinical study;
•BIVV003, our cell therapy product candidate for the treatment of sickle cell disease, or SCD, is currently being evaluated in our Phase 1/2 PRECIZN-1 clinical study. We are developing BIVV003 with our collaborator Sanofi; and
•ST-400, our cell therapy product candidate for the treatment of transfusion dependent beta thalassemia, is currently being evaluated in our Phase 1/2 Thales clinical study. We are developing ST-400 with our collaborator Sanofi.
In addition, we expect to initiate clinical studies for two additional product candidates in 2021:
•TX200, our wholly-owned Chimeric Antigen Receptor, or CAR, engineered regulatory T cell, or CAR-Treg, cell therapy product candidate for the treatment of HLA-A2 mismatched kidney transplant rejection; and
•KITE-037, our allogeneic anti-CD19 CAR-T cell therapy product candidate for the treatment of cancer. We are developing KITE-037 with our collaborator Kite, a wholly-owned subsidiary of Gilead Sciences, Inc.
Moreover, we are focusing our preclinical development in emerging areas for us including CAR-Treg cell therapies for autoimmune disorders and genome engineering for neurological diseases. Indications for our other preclinical programs include neurodevelopmental disorders, cancer, inflammatory bowel disease, or IBD, tauopathies such as Alzheimer’s and neurodegenerative diseases such as amyotrophic lateral sclerosis, or ALS, multiple sclerosis, or MS, and Huntington’s disease,
some of which we are developing with our collaborators Biogen, Kite, Novartis Institutes for BioMedical Research, Inc., or Novartis, Pfizer and Takeda.
Our multiple collaborations with biopharmaceutical companies bring us important financial and strategic benefits and reinforce the potential of our research and development efforts and our zinc finger protein, or ZFP, technology platform. They leverage our collaborators’ therapeutic
areas: inherited metabolic diseases, or IMDs, central nervous system diseases and
inflammatoryclinical expertise and
autoimmune diseases.commercial resources with the goal to bring our medicines more rapidly to patients. We believe these collaborations reflect the value of our ZFP technology platform and will potentially expand the addressable markets of our product candidates. To date, we have received approximately $815.0 million in upfront licensing fees, milestone payments and proceeds from sale of our common stock to collaborators and have the right to earn up to $7.0 billion in future milestone payments from our collaborations, in addition to potential product royalties.
Our Novel Science
We are a leader in the research and development of zinc finger proteins, or ZFPs, awhich are naturally occurring class ofsequence specific DNA binding proteins found in humans.humans and other species. We have used our knowledge and expertise to developdeveloped a proprietary synthetic ZFP technology platform with potential clinical utility in both(i) genome editing and gene regulation.genome regulation, which we refer to together as genome engineering and (ii) gene-edited cell therapy, which we refer to as cell therapy.
Our strategy is to translate our differentiated and versatile ZFP technology platform to product candidates with best- or first-in-class clinical potential. For example, ZFPs can be engineered to make zinc finger nucleases, or ZFNs, which are proteins that can be used to edit genomes by specifically modifymodifying DNA sequences by addingknocking in or knocking out specific genes, or genome editing, andselect genes. ZFPs can also be engineered to make ZFP transcription factors, or ZFP TFs,ZFP-TFs, which are proteins that can be used to increaseregulate genomes by selectively increasing or decreasedecreasing gene expression, or gene regulation. expression.
In the process of developing
this platform,these genome engineering technologies, we have
additionally accrued significant scientific, manufacturing and development capabilities,
andas well as related know-how, that are
generallybroadly applicable
into the
broader field of gene therapy,
andwhich we have
capitalized this knowledge into a conventionalused to develop our gene therapy
platform.Our Business Strategy
Our strategy isproduct candidates.
Finally, we have also leveraged our ZFP technology platform and technologies obtained through acquisitions to
maximize the valuebecome a leader in researching and
therapeutic use of our technology platforms. In certain therapeutic areas, we intend to capture the value of our proprietary genome editing and gene therapy products by forward integrating into manufacturing, development and commercial operations. In other therapeutic areas, we intend to partner with biopharmaceutical companies to develop products as appropriate. Decisions to partnerdeveloping CAR-Treg product candidates
or not will be based on our evaluation of the best way to bring new medicines to patients and based on our evaluation of our capacity to bring such products to commercial stage rapidly and efficiently on our own.2018 and Early 2019 Performance Highlights
In February 2018, together with Case Western Reserve University, we announced an $11.0 million grant from the National Institutes of Health for a planned study of gene-edited T cells designed to eradicate persistent HIV infection in patients receiving anti-retroviral therapy.
In February 2018, we entered into a global collaboration and license agreement with Kite Pharma, Inc., a wholly-owned subsidiary of Gilead Sciences, Inc., or Kite Pharma, for the research, development and commercialization of potential engineered cell therapies for cancer. We (i) received a $150.0 million upfront payment from Kite, (ii) are eligible to receive contingent
28
| development- and sales-based milestone payments that could total up to $3.01 billion if all the specified milestones set forth in this agreement are achieved, and (iii) will be entitled to receive escalating, tiered royalty payments with a percentage in the single digits based on potential future annual worldwide net sales of licensed products.
|
In April 2018, we completed an underwritten public offering of our common stock, in which 14,156,500 shares of our common stock were sold at a public offering price of $16.25 per share. The aggregate gross proceeds, before deducting the underwriting discounts and commissions and offering expenses, were approximately $230.0 million.
In May 2018, the U.S. Food and Drug Administration, or FDA, accepted the IND application for BIVV003, a gene-edited cell therapy candidate for the treatment of peopleautoimmune and inflammatory diseases in broad patient populations, including kidney transplant rejection, MS and IBD. CAR-Tregs are considered to have enhanced suppressive function over polyclonal regulatory T cells, or Tregs, due to the antigen-specificity introduced by the CAR.
Our In-House Manufacturing
We believe that our current and future in-house manufacturing capacity provides us a competitive advantage. We currently operate an in-house adeno-associated virus, or AAV, manufacturing facility in our Brisbane, California headquarters, and we are building cell therapy manufacturing facilities in Brisbane, California and Valbonne, France, which we expect to be operational by the end of 2021. Our manufacturing strategy is to provide greater flexibility, quality and control by building a balanced and necessary capacity achieved through our in-house manufacturing and contract manufacturing organization, or CMO, partnerships, investing in manufacturing processes and analytics and developing a strong supply chain.
2020 Performance Highlights
During 2020, we continued to advance the application and value of our technology platforms, as we work towards becoming a sustainable, fully integrated biopharmaceutical company. Our 2020 accomplishments included the following:
•New collaborations
◦We entered into a new collaboration with sickleNovartis for the development of gene regulation therapies to treat neurodevelopmental disorders.
◦We entered into a new collaboration with Biogen for the development of gene regulation therapies to treat neurological diseases.
•Clinical progress
◦We achieved clinical milestones under our collaborations with Pfizer related to the initiation of the Phase 3 AFFINE registrational trial for giroctocogene fitelparvovec, our first Phase 3 product candidate, and the completion of our research obligations under our genome engineering collaboration developing product candidates to treat amyotrophic lateral sclerosis, or ALS, and frontotemporal lobar degeneration.
◦We initiated our Phase 1/2 STAAR study evaluating our ST-920 gene therapy product candidate to treat Fabry disease.
◦We continued to prepare and seek the regulatory approvals for initiating our planned Phase 1/2 STEADFAST clinical study evaluating our TX200 CAR-Treg cell disease being developed by Bioverativ, Inc.,therapy product candidate to treat HLA-A2 mismatched kidney transplant rejection.
•Enhanced manufacturing capabilities
◦We brought online in 2020 an in-house AAV manufacturing facility in our collaboration partner.Brisbane, California headquarters and continued building cell therapy manufacturing facilities in Brisbane, California and Valbonne, France.
In July 2018,•Grew resources and infrastructure
◦We began 2020 with cash, cash equivalents and marketable securities of $384.3 million and finished 2020 with cash, cash equivalents and marketable securities of $692.0 million.
◦We continued to grow our personnel and infrastructure.
◦We achieved these 2020 accomplishments while successfully managing impacts to our business and operations from the global COVID-19 pandemic.
Objectives of Our Compensation Program
We are focused on translating groundbreaking science into genomic medicines with the potential to transform people’s lives using our platform technologies in gene therapy, cell therapy and genome engineering. To achieve this strategic business objective, we treated the first patientemphasize recruiting executives with significant industry or scientific experience, including in the Phase 1/2areas of clinical trial evaluatingSB-318 for MPS I.
In July 2018, we entered into agreements providing fordevelopment, medical affairs, product development, commercial planning and research innovation. This is a highly competitive industry, and our success depends upon our ability to attract and retain qualified executives through competitive compensation packages. The Compensation Committee administers the acquisition of TxCell, S.A., or TxCell, a leader in the emerging field of regulatory T cell, or Treg, development for immunological diseases, and in the fourth quarter of 2018, we completed the acquisition of 98.2% of the outstanding share capital and voting rights of TxCell for aggregate purchase consideration of approximately $80.4 million. We expect that the acquisition will accelerate our entry into the clinic with aCAR-Treg (Tregs genetically modified with a chimeric antigen reception, or CAR) therapy. In addition, we intend to use our ZFN gene editing technology to develop next-generation autologous and allogeneicCAR-Treg cell therapies for use in treating autoimmune diseases.
| • | | In December 2018, we treated the first patient in the Phase 1/2 clinical trial ofSB-FIX, ourin vivo genome editing therapy for hemophilia B.
|
In February 2019, the FDA accepted our IND application forST-920, a gene therapy candidate for Fabry disease.
| • | | In February 2019, we announced preliminary molecular and enzymatic evidence of editing of the human genomein vivo, as part of our interim results from the clinical trials evaluatingSB-318 and SB 913.
|
In February 2019, we announced our development of second generation, potentially more potent ZFN constructs designed to increase gene editing efficiency. The second-generation ZFNs are being manufactured with plans to initiate a clinical trial to evaluate second-generation ZFNs for SB-913 to treat MPS II in the second half of 2019 that we expect will enable a Phase 3 decisioncompensation programs for our MPS IIexecutive officers with this competitive environment and our need to attract and retain qualified executives in mind.
To this end, the primary objectives of our compensation program in 2020, and will allow us to define the next steps for the SB-318 and SB-FIX programs.are to:
Alignment of Executive Compensation Program with Business Strategy
Pharmaceutical
Biotechnology research, development and commercialization require sustained and focused effort over many years and
involvesinvolve a high degree of risk. As a consequence, the Compensation Committee believes our compensation program must balance long-term incentives that reward for the realization of our long-term strategic objectives with near-term compensation that rewards for the achievement of annual goals that further the attainment of our long-term objectives and align the interests of our executives with those of our stockholders.
The Compensation Committee generally believes that a formulaic or purely quantitative approach to executive compensation is not the best way to foster long-term success for us as a clinical-stage biotechnology company that is not yet profitable. Instead, the Compensation Committee sets annual performance objectives on which it believes our executive officers should focus during the year in order to achieve our business goals, including, for
2018,2020, aggressive
pre-established clinical, R&D,
manufacturing, business and corporate development objectives.
29
The Compensation Committee strives to create a positive relationship between its compensation program and our corporate performance and considers competitive market dynamics, the business environment in which the results were achieved and any unplanned positive or negative events when making compensation decisions. A significant portion of the total compensation opportunity for each of our named executive officers is directly related to our stock price and to other performance factors that measure our progress against our strategic objectives.
In order to assure that the compensation programs for our named executive officers remain competitive with peer companies, accomplish our
pay-for-performance objectives, and create rewards for the realization of our long-term strategic objectives, the Compensation Committee works with Radford to obtain the advice and market data needed to ensure that the compensation programs achieve these goals.
Key Features of Our Executive Compensation Program
| | | | | | | | |
What We Do | | What We Don’t Do |
✓ü Design executive compensation to align pay with performance ✓ü Emphasize performance-based compensation, with 100% of CEO’s cash incentive compensation based onpre-established corporate performance goals ✓ü Include a “double-trigger” change-in-control provision in all equity awards granted to our named executive officers ü Cap the cash incentive compensation plan payouts ✓ü Subject our program to independent review by the Compensation Committee’s independent compensation consultant | | X Allow repricing of stock options without stockholder approval X Offer significant perquisites or personal benefits to our named executive officers X Allow hedging or pledging of our securities by employees X Offer a defined benefit pension plan, deferred compensation plan or supplemental executive retirement plan |
How We Determine Executive Compensation
Role of the Compensation Committee and Executive Officers
The Compensation Committee assists the Board of Directors in fulfilling its fiduciary responsibilities with respect to the oversight of our affairs in the areas of our compensation plans, policies and programs, especially those regarding executive compensation and employee benefits.
The Compensation Committee’s responsibilities include, among other things,things: (i) periodically reviewing and approving a philosophy for the compensation of our executive officers and key employees; (ii) adopting, amending, terminatingadministering and administering the Company’sreviewing compensation plans and programs for our executive officers and key employees, including among others, equity plans, incentive plans, bonus plans, stock purchase plans, severance plans, pension and profit sharing plans and retirement plans; (iii) reviewing, consideringapproving executive employment contracts and severance and change in control arrangements; (iv) approving compensation programs applicable tonon-employee members of the Board of Directors; (iv) establishingBoard; (v) approving general compensation levels for our executive officers, (other thanwith the CEO) and key employees; (v) reviewing andexception of Dr. Macrae; (vi) evaluating the performance and leadership of Dr. Macrae and recommending to the CEO and determining the CEO’sBoard his total compensation package; (vi)(vii) reviewing with the CEODr. Macrae his evaluation of the performance of the executive officers (otherother than the CEO)him and of our key employees, and approving each such executive officer’s and key employees to approve theiremployee’s total compensation package; (vii)(viii) reviewing and monitoring management development plans and activities; (viii) reviewing with our Board our succession plan for the CEO and other executive officers; and (vii)plans; (ix) conducting risk assessment with respect toassessments of our compensation programs. programs; (x) considering stock ownership guidelines for directors and employees; and (xi) reviewing and discussing with management our Compensation Discussion and Analysis and preparing a report on executive compensation, each for inclusion in this Proxy Statement.
The Compensation Committee does not delegate any of its functions to others in determining executive compensation.
In making executive compensation determinations, the Compensation Committee considers recommendations from Dr. Macrae. In making his recommendations, Dr. Macrae receives input from our human
30
resources department and from the individuals who manage the other executive officers. While Dr. Macrae discusses his recommendations for the other executive officers with the Compensation Committee, he does not participate in the deliberations or determination of his own compensation. Members of our finance, human resources and legal departments mayperiodically attend Compensation Committee meetings from time to time.
Objectives of Our Compensation Program
We are focused on translating groundbreaking science into genomic medicines with the potential to transform people’s lives using our platform technologies in genome editing, gene therapy, gene regulation and cell therapy. To achieve this strategic business objective, we emphasize recruiting executives with significant industry or scientific experience, including in the areas of clinical development, medical affairs, product development, commercial planning and research innovation. This is a highly competitive industry and our success depends upon our ability to attract and retain qualified executives through competitive compensation packages. The Compensation Committee administers the compensation programs for our executive officers with this competitive environment and need to attract and retain qualified executives in mind.
To this end, the primary objectives of our compensation program are to:

meetings.
Factors Used in Determining Executive Compensation
The Compensation Committee sets the compensation of our executive officers at levels that the Compensation Committee determines to be competitive and appropriate for each named executive officer, using the Compensation Committee’s professional experience and judgment. The Compensation Committee does not use a formulaic approach to setting executive pay, but rather it considers multiple relevant factors, which may vary from year to year. The figure below reflects the factors the Compensation Committee generally considers in determining and approving the amount, form and mix of pay for our named executive officers.

31
Role of Independent Compensation Consultant
The Compensation Committee retained the services of Radford in order to
(i)(1) review the peer group selection criteria and specific peer companies, (2) assess compensation levels and mix of elements for our executive officers and vice presidents for
2018, (ii) review the peer group selection criteria and specific peer companies, (iii)2020, (3) advise the Compensation Committee on executive compensation and governance trends based on peer group trends and market practices (including severance arrangements), and
(iv)(4) advise the Compensation Committee on compensation programs applicable to our
non-employee directors.
In 2018, Radford also
providedprovides the Compensation Committee with comparative
market data in connection with setting the compensation for
Ms. Turner and Mr. Boissel when each individual joined us.new executives. The Compensation Committee determined that Radford was independent using criteria established by Nasdaq for evaluating the independence of advisors to the Compensation Committee.
For purposes of measuring the competitive positioning of our compensation packages
peer companies are generally selected byfor 2020, in September 2019, the Compensation Committee
selected our peer companies primarily using the following criteria: (i) companies that fall within a specific market capitalization range of approximately
one-third to three times our market capitalization at the time of the peer evaluation ($
250500 million to
$2.5$5.0 billion at the time we selected our
20182020 peer group); (ii)
biotechnologybiopharmaceutical companies in the
pre-commercial Phase 2 or Phase 3 stage of development; and (iii) companies with
50200 to
500800 employees. In addition, we prefer companies that are headquartered in
core biotechnology hubs, particularly within the San Francisco Bay Area
and other California markets, where we both recruit and may lose executive talent. Because the biotechnology industry is a dynamic industry, the comparator group used by the Compensation Committee to measure the competitive positioning of our compensation packages is periodically updated to ensure that companies continue to meet the established criteria. At the time the peer group was established, our
30-day market capitalization was comparable to the
63th33rd percentile of the peers, validating the reasonableness of the peer group.
The selected comparable companies for purposes of making compensation decisions for
2018,2020, or the
20182020 peer group, were as
follows:follows, with new additions to the 2020 peer group in bold: | | | | | | | | |
| Acceleron Pharma | | Insmed |
Achaogen | | IntelliaFate Therapeutics |
| Aimmune Therapeutics | | Intellia Therapeutics |
| AnaptysBio | | Iovance Biotherapeutics |
| Arena Pharmaceuticals | | MacroGenics |
| Arrowhead Pharmaceuticals | | Mirati Therapeutics |
| Atara Biotherapeutics | | MyoKardia |
| Audentes Therapeutics | | MannKindOdonate Therapeutics |
bluebird bio | | Peregrine Pharmaceuticals |
Celldex TherapeuticsChemoCentryx | | REGENXBIO |
ChemoCentryxCRISPR Therapeutics | | Rigel PharmaceuticalsRevance Therapeutics |
| Cytokinetics | | Spark Therapeutics |
Dynavax TechnologiesDenali Therapeutics | | Ultragenyx Pharmaceutical |
| Editas Medicine | | Versartis |
Global Blood Therapeutics | | Voyager Therapeutics |
ImmunoGen | | Xencor |
The following companies were part of the selected comparable companies for purposes of making compensation decisions for 2019 but were excluded from the 2020 peer group as they no longer complied with the updated selection criteria: bluebird bio, Dynavax Technologies, Global Blood Therapeutics, Immunogen and Rigel Pharmaceuticals.
When setting pay, the Compensation Committee examines the market data derived by Radford from the peer group that ranges from the
25th25th to the
75th75th percentiles. We do not have a set formula or benchmark for
pay, but ratherpay. Instead, the Compensation Committee references the market data as one input before making position-specific compensation decisions. We generally seek to manage compensation
between the 50th and 75th percentiles ofwithin the peer group
data,range, if warranted by performance and other factors the Compensation Committee considers relevant in making specific
pay decisions. A specific executive officer may be paid below or above the target range based on individual performance, experience, skills and the importance of the executive’s position to us. The
32
Compensation Committee considers this positioningapproach to determining pay appropriate given that we are basedoperate in the highly competitive San Francisco Bay Area talent market, where there is a high concentration ofthe particularly intense competition for executive talent competitors creating competitive pressure that requires us to ensure that our overall compensation program is competitive to attract and retain strong leadership for our long-term success based on performance.
2018
2020 Advisory Vote on Executive Compensation
At the
2018 Annual Meeting,2020 annual meeting of stockholders, we conducted our
thirdfifth advisory vote on executive compensation. At such meeting, approximately
96%91% of the votes cast on the advisory vote proposal were supportive of our named executive officer compensation program as disclosed in our proxy statement.
The Compensation Committee reviewed the advisory vote results in the context of our overall compensation philosophy and programs, and based on the level of support, determined that no significant changes to our compensation policies and programs were necessary. The Compensation Committee will continue to consider the results from future stockholder advisory votes on executive compensation and other relevant market developments affecting executive officer compensation in order to determine whether any subsequent changes to our executive compensation programs and policies would be warranted to reflect any stockholder concerns reflected in those advisory votes or to address market developments.
Design of the Executive Compensation Program and Key Components
Target Total Compensation
As discussed above, the development and commercialization of
pharmaceuticalbiotechnology products involve a high degree of risk. The Compensation Committee regularly evaluates the relative importance of equity and cash components of total compensation when determining the level and structure of annual pay based on the business priorities established by the Board of Directors. Target total compensation for
2018,2020, as shown below for Dr. Macrae, reflects annual base salary, annual target cash incentive compensation and the grant date fair value of stock options
and RSU awards granted during the year (as such stock options
and RSU awards are reported in the Summary Compensation Table).
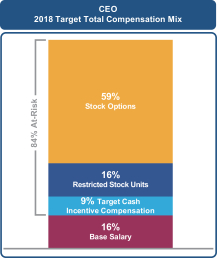
33
The Compensation Committee at least annually assesses the allocation of equity awards between stock options and RSU awards in connection with a review of our outstanding shares, our stock price and comparative market data of peer companies.
Key Elements of Our Executive Compensation Program
| | | | | | | | |
Component | | Key Features | | Purpose |
| | |
Base Salary | | •Annual increases are not guaranteed •Amounts are reviewed and determined annually (or at the time of a change in the executive’s title or position during the year) •Amounts determined based on market data, individual performance, experience, skills and the importance of the executive’s position | | •Enables us to attract and retain skilled and experienced executives and to provide a level of economic security for executives from year to year •Provides fixed level of compensation that is competitive within our industry and geographic area |
|
|
|
| | |
| Cash Incentive Compensation | | •Cash compensation under the Sangamo Therapeutics, Inc. Amended and Restated Incentive Compensation Plan, or the Incentive Plan, which is dependent upon achievement of performance objectives •Target amounts are reviewed and determined annually (or at the time of a change in the executive’s title or position during the year) and expressed as a percentage of base salary •Actual amounts paid are based on the extent corporate objectives are attained as determined by the Compensation Committee, and for executive officers other than Dr. Macrae, on the achievement of pre-established individual goals that align with our overall priorities | | •Provides each executive officerofficers with a direct financial incentive in the form of cash incentive compensation tied to our achievement of aggressive pre-established clinical, research and development, business and corporate developmentperformance goals for the year based on our annual operating plan •Aligns pay structure and mix with, and ensures that our total cash compensation is within, competitive norms within the peer group and more broadly in the sector •Aligns a portion of the executive’s cash compensation to specificpre-established corporate and individual goal achievement to reinforce our performance-based culture |
|
|
34
| | | | |
Component
| | Key Features
| | Purpose
|
| | |
| Equity Compensation | | •Generally in the form of stock options and/or RSUs which are “at-risk” “at-risk” because the realized value is dependent upon our stock priceprice. We generally allocate total equity value in a 2:1 ratio between stock options and RSUs. Vesting schedules have been devised with a goal of retention and long-term accountability •The exercise price for stock options is equal to the closing price of our stock on the option grant date. 25% of the option shares vest and become exercisable on the one-year anniversary of the option grant date and the remaining option shares vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the option grant date, subject to continuous service through each applicable vesting date •Each RSU provides the recipient with one share of our common stock at a designated issue date following the vesting of that unit, without the payment of an exercise price or other cash consideration for the issued shareshare. 1/3rd of the shares vest in three equal annual installments over the three-year period measured from the grant date, subject to continuous service through each applicable vesting date •Executives are eligible for equity awards in connection with annual performance reviews and at time of hire | | •Provides long-term incentives that align the interests of our work force with the achievement of our long-term vision to develop and commercialize pharmaceutical products which occurs over time •Given the time periods involved in pharmaceuticalbiotechnology development, we believe that long-term incentives are critical to our success, to provide long-term focus,as they aid in retention, and mitigate short-term risk taking to realize gains in the near term that creates risk in theand instill a long-term focus on maximizing stockholder value •We believe that a mix of stock options and restricted stock unitsRSUs reinforces the long-term nature of our business and alignment with our stock holdersstockholders by rewarding for improvements in stock price over a period of time. The company issuestime; we issue stock options to reward for future performance and appreciation while providing restricted stock unitsRSUs to manage the natural market volatilities forof a development stageclinical-stage company, provide retention incentives during the vesting period and to reinforce a culture of ownership.ownership •By granting RSUs, the companywe can also reduce the dilutive effect of the equity incentive awards, which benefits our stockholders over time |
|
|
|
2018
2020 Compensation Decisions
2018
2020 Base SalaryIn general,
We determine increases to the base
salariessalary of all employees annually as part of our
named executive officers for 2018 were between the 25th and 75th percentiles of the market data derivedannual performance review process. These increases generally range from
the peer group.0-5%.
In
accordance with our practice, in December
20172019, we conducted annual performance reviews for
20172019 for the named executive officers. In early
2018,2020, the Compensation Committee considered these reviews,
our 2017 performance,role criticality and advice from Radford and
competitivecomparative data from
theour peer group and determined to increase base salaries as set forth below. The Compensation Committee specifically considered Dr. Macrae’s responsibility for the Company’s achievement of its corporate objectives;
Ms. Yi’s role asMr. Lee’s work in strategizing external communications and meeting with investors, his effective oversight of facilities and IT, and his preparation for 2020 investor conferences; Mr. Loeb’s successfully building out a
business partnerhigh-performing legal department, overseeing the Company’s corporate governance practices and
that extends35
beyond her function, leadership of a high performing team and leadership of transformative transactions; and Dr. Conner’s rolepolicies, supporting the Board and all of its committees, participating in and supporting licensing and collaboration transactions, and updating internal legal systems and infrastructure; Dr. Ramelmeier’s leadership in growing our technical operations capacities and skills, his role in leading the successful completion and articulation of the manufacturing roadmap, and his oversight of our successful global integration of manufacturing and R&D priorities and programs; and Mr. Boissel’s business development organizationleadership for the career developmentBiogen and Novartis deals, his accomplishments as our interim principal financial officer, his leadership in providing cross-functional support for IT, facilities and alliance management, and his support of membersthe corporate integration of his team.
| | | | | | | | | | |
Name | | 2018
Base Salary ($) | | | 2017
Base Salary ($) | | Percent
Increase | |
Alexander D. Macrae | | | 636,480 | | | 612,000 | | | 4% | |
Kathy Y. Yi | | | 404,833 | | | 350,000 | | | 16% | |
Heather D. Turner (1) | | | 400,000 | | | — | | | N/A | |
Edward R. Conner, M.D. | | | 408,000 | | | 400,000 | | | 2% | |
| (1) | Effective as of February 12, 2018 upon commencement of Ms. Turner’s employment as Senior Vice President, General Counsel and Secretary after taking into consideration her compensation at her prior employer, competitive positioning based on the market data derived by Radford from the peer group that ranges from the 50th to the 75th percentiles and internal positioning relative to the Company’s other executive officers. Ms. Turner resigned from the Company effective March 15, 2019.
|
Sangamo Therapeutics France, S.A.S., or Sangamo France. In general, the base salaries of our named executive officers for 2020 were between the 50th and 75th percentiles of the market data derived from the peer group.
| | | | | | | | | | | | | | | | | | | | |
| Name | | 2020 Annual Base Salary ($) | | 2019 Annual Base Salary ($) | | Percent
Increase |
| Alexander D. Macrae | | 681,797 | | | 661,939 | | | 3% |
| Sung H. Lee (1) | | 438,262 | | | 435,000 | | | 1.5% |
| Gary H. Loeb (2) | | 406,000 | | | 400,000 | | | 1.5% |
| D. Mark McClung (3) | | 415,000 | | | — | | | NA |
| R. Andrew Ramelmeier | | 416,000 | | | 400,000 | | | 4% |
| Stéphane Boissel (4) | | 502,944 | | | 483,600 | | | 4% |
___________________
(1)Mr. Lee’s base salary percentage increase was prorated from 3% to reflect that he commenced employment with us in October 2019. He resigned effective February 1, 2021.
(2)Mr. Loeb’s base salary percentage increase was prorated from 3% to reflect that he commenced employment with us in July 2019.
(3)Mr. McClung commenced employment with us on May 1, 2020.
(4)Mr. Boissel resigned effective July 31, 2020.
In connection with
theMr. McClung’s commencement of
his employment
with the Company, after taking into consideration his compensation at his prior employer, competitive positioning based on the market data derived by Radford from the peer group that ranges from the
50th25th to the
75th75th percentiles and internal positioning relative to the Company’s other executive officers, the Compensation Committee determined that Mr.
Boissel’sMcClung’s annual base salary would be
$480,000.2018$415,000.
2020 Cash Incentive Compensation
In
March 2018,January 2020, we adopted a
2018 Executive Officer Bonus Program, or the 20182020 Cash Incentive Compensation Program under the Incentive Plan that provides for annual cash incentive compensation to reward executive officers based on our achievement of
specificpre-established corporate goals and,
for all executive officers other than Dr. Macrae, their achievement of individual performance goals. The
objectivesprogram included several aggressive goals to encourage our employees to achieve more than target performance and
weightingswas designed with a potential of earning 150 points, with the expectation that
were approved byachievement of 100 points (i.e., two-thirds achievement) would represent target performance and that the total points would correlate to earning an equivalent percentage of the bonus target such that 100 points would result in a 100% annual cash incentive compensation multiplier relative to the bonus target with respect to corporate performance. In December 2019, the Board
of Directors in March 2018 under the 2018 Cash Incentive Compensation Programapproved our 2020 corporate goals and accompanying potential points, which are
describedsummarized in the chart below.
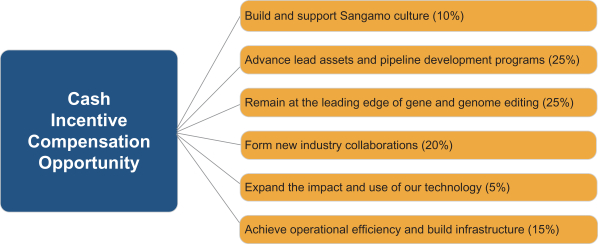
For
2018,2020, the target cash incentive compensation for Dr. Macrae was
equal to 60% of
his base salary
for 2020, and for
each of our other named executive officers,
(other than Mr. Boissel, who did not receive any cash incentive compensation under the 2018 Cash Incentive Compensation Program given that his full-time employment with us commenced in January 2019), it was
35%equal to 40% of
their respective base
salary.salaries. Dr. Macrae’s cash incentive compensation opportunity was based solely on corporate performance and each of our other named executive officers’ cash incentive compensation opportunity was allocated between corporate and individual performance
(80%(90% and
20%10%, respectively).
36
In February 2019,December 2020, the Compensation CommitteeBoard determined that our 2018we earned 125 points out of a total maximum possible 150 points, resulting in a 125% multiplier with respect to corporate objectives were attained at 110%,performance, as further described below.
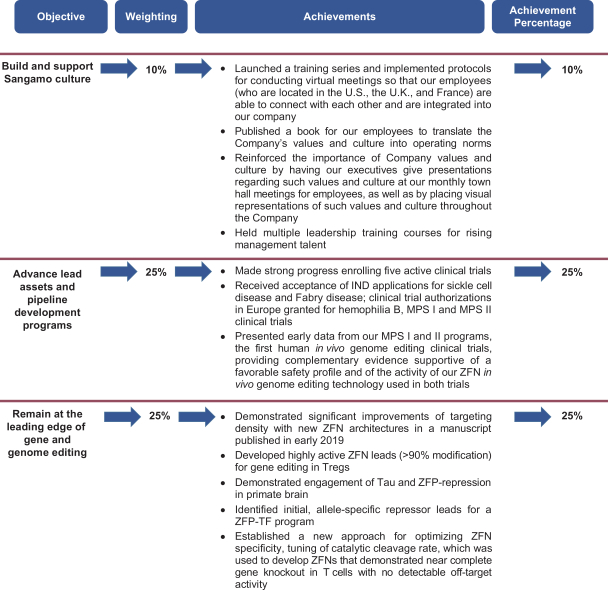
37
The 125% multiplier was used in part in calculating annual cash incentive payments for all eligible employees, including our executive officers. The Board did not adjust the corporate objectives to reflect impacts to our business or operations of the global COVID-19 pandemic. | | | | | | | | | | | |
| Objective | Potential Points | Achievements | Points Achieved |
| Advance lead assets programs | 15 | Fabry: Progress on Phase 1/2 STAAR study TX200: Site approvals for the planned Phase 1/2 STEADFAST study
| 10 |
| Advance partnered programs in line with contractual terms | 12 | Pfizer - SB525: Investigational new drug, or IND, transferred to Pfizer Pfizer - C9ORF: Completion of research activities Kite/Gilead - CD19: Deliverables completed for IND submission Collaborations: Supported all Phase 1/2 activities and Phase 3 readiness activities as defined by agreement with collaborator
| 8 |
| Progress new business opportunities | 50 | New collaboration agreements with Biogen and Novartis
| 45 |
| Build sustainable infrastructure and capabilities to progress Sangamo proprietary assets toward commercialization | 15 | Brought online in-house manufacturing Functional 3-year roadmaps aligned with LRP in place Expanded project portfolio management capability Functional talent onboarding plans, talent reviews and succession plans Developed enterprise compliance program and quality management systems
| 13 |
| Maintain approximately two years of cash, cash equivalents and marketable securities at year end | 15 | Began 2020 with cash, cash equivalents and marketable securities of $384.3 million and finished 2020 with cash, cash equivalents and marketable securities of $692.0 million
| 14 |
| Build pipeline and advance assets toward IND | 25 | Identified new pipeline targets Advanced pre-clinical programs Continued building cell therapy manufacturing facilities for opening in 2021
| 20 |
| | | | | | | | | | | |
| Objective | Potential Points | Achievements | Points Achieved |
| Remain at the leading edge of gene editing and delivery technologies and explore innovative approaches in genomic medicine | 10 | Increased throughput of gene assembly and screening New functions: Developed at least one novel ZFP functionality to the point of sufficient performance for clinical use Delivery: Completed current AAV serotype development studies with test of new variant AAVs Developed Engineered Treg Platform: Established CAR screening platform Maintained scientific leadership: Met high-impact publication targets and present at key conferences
| 8 |
| Build Sangamo culture and support employees based on our core values | 8 | Employment engagement survey: Achieved result above 2019 baseline Mindset: Created a continuous improvement community of practice and a value creation baseline Culture: Fostered a culture that embraces the voice of patients
| 7 |
| TOTAL | 150 | | 125 |
In addition, in
February 2019,January 2020, the Compensation Committee approved individual achievement percentages for our named executive officers (other than Dr. Macrae, whose cash incentive compensation opportunity was tied solely to achievement of the
20182020 corporate objectives) based on Dr. Macrae’s assessment of and recommendations regarding each executive’s accomplishments against
pre-established individual objectives, as further described below. As described above, Dr. Macrae does not have individual goals separate from our corporate objectives. For our other named executive officers, the total cash incentive compensation payout for
20182020 was based on a weighting of
80%90% corporate and
20%10% individual goals. Dr. Macrae recommends individual goals for each other named executive officer, which are aligned with our business strategy and linked with corporate goals, and our Compensation Committee approves these goals. The
20182020 individual goals for the named executive officers include those listed below. These specific goals were in addition to the general responsibilities each officer had for managing his or her respective functional operational area. Based on the recommendation of Dr. Macrae, as well as the observations by Compensation Committee members of these executive officers and its own assessment of each individual’s effectiveness, the Compensation Committee determined the level of achievement of each named executive officer’s individual performance goals as follows:
Ms. Yi –•Mr. Lee — The Compensation Committee determined that with respectMr. Lee was successful in leading the financial organization, facilities and IT for 2020. In addition, Mr. Lee successfully chaired our COVID-19 task force to help navigate our response to the objectives of keeping compliantpandemic. Mr. Lee standardized our analysis and managing all internalreporting for our financial positions, helped lead efforts to evolve our strategy in product development, optimized communication with investors and externalanalysts and led a diversified and growing financial requirements through effective controls and processes, being a useful business partner as ateam.
go-to• function, and going beyond her function and leading a high performing team, Ms. Yi achieved all of her individual objectives by executing her finance responsibilities, upgrading processes, invigorating and integrating her team as well as other internal departments, supporting and leading transformative transactions, and playing a key role in the implementation of ERP. In addition, Ms. Yi led the design and renovation of the Brisbane facility and the two site integration on time and on budget.Mr. Loeb — The Compensation Committee determined that Ms. YiMr. Loeb achieved 110% of herhis individual objectives. Mr. Loeb’s key accomplishments in 2020 included: developing a company-wide coordinated compliance program; supporting business development collaborations and transactions; strategizing with respect to intellectual property infrastructure and assessments; serving as interim head of human resources for approximately half of the year; and supporting the Board in corporate governance matters and in his capacity as Corporate Secretary.
38
Ms. Turner –•Mr. McClung — The Compensation Committee determined that with respect to the objectives related to leadership and management of the legal and human resources teams and acting as General Counsel to the CEO and executive team and secretary to the Board of Directors, Ms. TurnerMr. McClung achieved all of her individual objectives by managing meetings of the Board of Directors, surpassing her legal role by becoming involved in our business strategy, supporting and leading transformative transactions (including leading the negotiation process in the TxCell acquisition), leading her department, and supporting the development and management of other departments. In addition, Ms. Turner transformed both the legal and human resources departments, hiring excellent leaders in both departments and initiating a series of process improvements to create modern professional functions. The Compensation Committee determined that Ms. Turner achieved 110% of her individual objectives.
Dr. Conner – The Compensation Committee determined that with respect to the objectives of supporting the development organization, ensuring data quality and Good Clinical Practices, or GCP, compliant study conduct and data collection, raising the visibility of clinical development within our company, and supporting the career development of members of his team, Dr. Conner achieved all100% of his individual objectives by establishing a clinical group that is on track to meet its milestones and objectives, establishing clinical development plans, performing clinical assessments for pipeline projects, leading clinical contributions to IND applications, raising the visibility and successes of his department, and supporting the development andobjectives. Mr. McClung’s key accomplishments in 2020 included: successfully overseeing management of his team.business development, alliance management, government affairs, patient advocacy, communications and product strategy; developing the evolution of our corporate strategy and, working with Communications and IR, communicating the updated strategy to key internal and external stakeholders. Mr. McClung also spearheaded efforts to support our collaborations and optimize the new product planning assessments. In addition, Mr. McClung has served as the executive sponsor of our diversity, equity and inclusion initiatives.
•Dr. Ramelmeier — The Compensation Committee determined that Dr. ConnerRamelmeier achieved 100% of his individual objectives. Dr. Ramelmeier’s key accomplishments in 2020 included: building out GMP manufacturing capabilities internally; strategizing and leading with respect to build-out of manufacturing at Sangamo France; hiring additional capabilities within the Technical Operations team at all levels; and working with third-party manufacturers to improve processes and technology transfers.
The table below sets forth the target and actual cash incentive compensation for our named executive
officers: | | | | | | | | | | |
Name | | Target Amount ($) | | | Actual Amount ($) | | | % of Target |
Alexander D. Macrae | | | 381,888 | | | | 420,077 | | | 110% |
Kathy Y. Yi | | | 141,692 | | | | 155,861 | | | 110% |
Heather D. Turner(1) | | | 128,333 | | | | 141,166 | | | 110% |
Edward R. Conner, M.D. | | | 142,800 | | | | 154,224 | | | 108% |
| (1) | Ms. Turner’s target and actual cash incentive compensation amounts were prorated to reflect that she commenced employment with us in February 2018.
|
2018officers for 2020:
| | | | | | | | | | | | | | | | | | | | |
| Name | | Target Amount ($) | | Actual Amount ($) | | % of Target |
| Alexander D. Macrae | | 409,078 | | | 511,348 | | | 125% |
| Sung H. Lee (1) | | 175,305 | | | 215,000 | | | 123% |
| Gary H. Loeb | | 162,400 | | | 200,564 | | | 124% |
| D. Mark McClung (2) | | 110,667 | | | 136,245 | | | 123% |
| R. Andrew Ramelmeier | | 166,400 | | | 203,840 | | | 123% |
| Stéphane Boissel (3) | | 201,178 | | | — | | | — |
___________________
(1)Mr. Lee resigned effective February 1, 2021. On January 22, 2021, we entered into a letter agreement with Mr. Lee, pursuant to which Mr. Lee agreed to provide services to us in an independent contractor role for the period from February 2, 2021 through February 26, 2021. Mr. Lee’s compensation for this independent contractor role consisted solely of a payment of $215,000, payable in one lump sum on February 26, 2021, which was deemed to be the annual cash incentive compensation payable to him under the Incentive Plan for 2020.
(2)Mr. McClung’s target and actual cash incentive compensation amounts were prorated to reflect that he commenced employment with us on May 1, 2020.
(3)Mr. Boissel resigned effective July 31, 2020 and thus was not eligible for any annual cash incentive compensation.
Our current practice is to make equity grants at the beginning of the fiscal year after our operating plan has been approved by the Board of Directors and the performance in the prior fiscal year completed. Accordingly, the Compensation Committee approved the grant of stock options and
restricted stock unitRSU awards to Dr. Macrae,
Ms. Yi,Mr. Lee, Mr. Loeb, Dr. Ramelmeier and
Dr. ConnerMr. Boissel in January
2018.2020.
The Compensation Committee considered the approach used when determining the form and size of prior grants, the size of
20172019 equity grants, the percent of common stock outstanding as well as grant date
fair value, to determine the overall range for setting the pool of shares available for grants to executive officers and other employees in
20182020 and the size of each executive’s grant.
In 2018,The Compensation Committee also assessed the
percentallocation of
commonequity awards between stock
options and RSU awards in connection with a review of our outstanding
carried more weight due to the depressedshares, our stock price
in lightand comparative market data of
the objective of managing overall dilution for stockholders.peer companies. In general, the equity compensation of our named executive officers for
20182020 fell within the competitive range of the
50th25th to
75th75th percentiles based on the market data provided by Radford.
In connection with
herMr. McClung’s commencement of employment
in February 2018,with the Company, after taking into consideration
her compensation at her prior employer and competitive positioning based on the market data derived by Radford from the peer group,
that ranges frominternal peer comparisons and the
50thcompensation required to attract him to the
75th percentiles, Ms. Turnerrole with the Company, Mr. McClung was
a granted a stock option to
39
acquire 200,000 shares of common stock with an exercise price per share equal to the closing price per share on the grant date. The stock option is subject to a four-year vesting schedule, with 25% of the shares subject to the option vesting 12 months from the grant date and the remainder vesting in equal monthly installments for 36 months thereafter, provided that Mr. McClung remains a full-time employee through each vesting date. In addition, Mr. McClung was granted a RSU award covering 100,000 shares of common stock. Mr. McClung’s RSU award is subject to a three-year vesting schedule, with one-third of the shares subject to the RSU award vesting in a series of three successive equal installments upon Ms. Turner’s completion of oneeach year of service measured from the date Ms. Turner commenced employment with us and the balance of the shares vesting monthly thereafter for the next three years
In connection with his commencement of employment in October 2018, after taking into consideration his compensation at his prior employer and competitive positioning based on the market data derived by Radford from the peer group that ranges from the 50th to the 75th percentiles, Mr. Boissel was a granted a stock option to acquire 215,000 shares of common stock with an exercise price per share equal to the closing price per share on the grant date. Such stock option is subject to a four-year vesting schedule with 25% of the shares subject to the option vesting upon Mr. Boissel’s completion of one year of service measured from the option grant date and the balance of the shares vesting monthly thereafter for the next three years. Further, in connection with our acquisition of TxCell, Mr. Boissel, as an employee of TxCell, was granted a stock option under the 2018 Plan to acquire 68,523 shares of common stock with an exercise price equal to the closing price per share on the grant date. Such stock option is subject to a four-year vesting schedule with 25% of the shares subject to the option vesting upon Mr. Boissel’s completion of one year of service measured from the option grant date and the balance of the shares vesting monthly thereafter for the next three years.
Other Elements of Executive Compensation Program
The remaining elements of our executive compensation program, like our broader employee compensation programs, are intended to make our overall compensation program competitive with those of our peer companies and all of the remaining elements of our executive compensation program (401(k) Plan; Medical, Dental, and Vision Plans; Life and Disability Insurance; and
our 2010 Employee Stock Purchase Plan, as amended, or the
Purchase Plan)2020 ESPP) are available to all of our employees.
With respect to the perquisites and other benefits provided to
In 2020, Mr.
Boissel as discussed in the “Summary Compensation Table,” includingMcClung was granted a
car allowance and premiums for an unemployment insurance contract, as well as a transaction$150,000 sign-on bonus
awarded in connection with
our acquisitionthe commencement of
a controlling interesthis employment with the Company in
TxCell andMay 2020. In determining the size of Mr.
Boissel’s 2018 target incentiveMcClung’s sign-on bonus,
such amounts were approved and paid by TxCell and will not be providedthe Compensation Committee took into account several factors, including the need for Mr. McClung to relocate from Southern California to Northern California in connection with this role. If Mr. McClung’s employment with us terminates within one year of his appointment, either by the Company
going forward.for cause or by him for good reason, Mr. McClung’s sign-on bonus will be subject to repayment.
In December 2019, Dr. Ramelmeier was granted a $100,000 one-time special retention bonus to recognize the critical importance of his leadership and experience in advancing our manufacturing capabilities, especially as the Company works
toward completing the buildout of its manufacturing facilities in Brisbane and Valbonne. Dr. Ramelmeier’s special cash bonus is scheduled to be paid in four equal installments between July 2020 and December 2021, subject to Dr. Ramelmeier’s continued full-time employment with the Company through and including each payment date.
Additional Compensation Information
In March 2017, the Compensation Committee adopted an Executive Severance Plan, or the Severance Plan, to provide severance benefits to certain of our executive officers and other key employees, including our named executive officers, whose employment terminates under certain prescribed circumstances. In February 2019, the Compensation Committee approved an Amended and Restated Executive Severance Plan, or the Amended Severance Plan, to increase certain severance payments, as described below under “—Employment Contracts
and Separation and Change in Control Arrangements—Executive Severance Plan.” The Amended Severance Plan provides financial protection against the potential loss of employment in designated circumstances, which the Compensation Committee believes will allow the executives to focus attention on the best interests of the stockholders, without undue concern as to an executive’s own financial situation. The Compensation Committee has been advised by its independent compensation consultant that the benefits provided under these agreements are consistent with peer company and broader market practices and are fair and reasonable.
Mr. Boissel’s employment agreement provided that he was eligible to receive certain severance benefits under the Amended Severance Plan. Mr. Boissel resigned effective July 31, 2020 and pursuant to a Separation Agreement is receiving severance benefits of $628,680 over a 15-month period and is eligible for reimbursement of health care coverage costs under COBRA for 15 months. Mr. Boissel also received a $25,000 payment for consulting services rendered to the Company subsequent to his termination.
Tax and Accounting Effects of Executive Compensation
Under Section 162(m) of the
Internal Revenue Code,
of 1986, as amended, or
the Code,Section 162(m), compensation paid to
any publicly held corporation’seach of the Company’s “covered employees” that exceeds $1 million per taxable year
for any covered employee is generally
non-deductible.40
Prior to non-deductible unless the enactment ofcompensation qualifies for (i) certain grandfathered exceptions (including the Tax Cuts and Jobs Act, Section 162(m) of the Code, or Section 162(m), provided a performance-based compensation exception, pursuant to which the deduction limit under Section 162(m) did not apply to any compensation that qualified as “performance-based compensation” under Section 162(m). Pursuant to the Tax Cuts and Jobs Act, the performance-based compensation exception under Section 162(m) was repealed with respect to taxable years beginning after December 31, 2017, except thatexception) for certain transition relief is provided for compensation paid pursuant to a written binding contract which was in effect on November 2, 2017 and which is not materially modified in any material respect on or after such date.
Compensation paid to each ofdate or (ii) the Company’s “covered employees” in excess of $1 million per taxable year generally will not be deductible unless it qualifiesreliance period exception for the performance-based compensation exception under Section 162(m) pursuant to the transition relief described above. Because of certain ambiguities and uncertainties as to the application and interpretation of Section 162(m), as well as other factors beyond the control of the Compensation Committee, no assurance can be given that any compensation paid by the Company will be eligible for such transition relief and be deductible by the Company in the future. corporations that became publicly held on or before December 20, 2019.
Although the Compensation Committee will continue to consider tax implications as one factor in determining executive compensation, the Compensation Committee also looks at other factors in making its decisions and retains the flexibility to provide compensation for the Company’s named executive officers in a manner consistent with the goals of the Company’s executive compensation program and the best interests of the Company and its stockholders, which may include providing for compensation that is not deductible by the Company due to the deduction limit under Section 162(m). The Compensation Committee also retains the flexibility to modify compensation that was initially intended to be exempt from the deduction limit under Section 162(m) if it determines that such modifications are consistent with the Company’s business needs.
The Compensation Committee also considers the impact of Section 409A of the Code, and in general, our executive plans and programs are designed to comply with the requirements of that section so as to avoid possible adverse tax consequences that may result from
non-compliance.The accounting impact of our compensation programs is one of many factors that the Compensation Committee considers in determining the structure and size of our executive compensation programs. In general, the Company accounts for equity compensation paid to our employees under ASC 718, which requires us to estimate and record an expense over the service period of the equity award, and our cash compensation is recorded as an expense at the time the obligation is accrued.
Equity awards are generally granted to employees, including executives, when they are hired, and employees are eligible for additional equity awards in connection with annual performance reviews each year.
As described above, annual equity grants were historically made in December of each fiscal year; however, in 2016, the Compensation Committee determined that it would be a better business and governance practice to make grants at the beginning of the fiscal year after our operating plan has been approved by the Board of Directors and the performance in the prior fiscal year has completed. In 2018, the Compensation Committee determined that, with respect to equity awards granted to
any new
employee,employees, such awards would be granted on the
25th25th day of the month (or if such
25th day is not a trading day, the immediately preceding trading
day of that month)day) immediately following the month of the employee’s hire date; and with respect to annual equity awards
granted to existing employees, such awards
willwould be granted on the
25th25th day of February (or if such
25th day is not a trading day, the immediately preceding trading
day in February)day), provided the Compensation Committee approves the terms of the grants before such date.
In 2020, the Compensation Committee determined that, with respect to equity awards granted to new employees, starting in January 2021, such awards would be granted on the second Friday of each month (or if such day is not a trading day, the immediately preceding trading day) for all new employees whose hire date is prior to this grant date. Because these grant dates are pre-established, the timing of the release of material
non-public information does not affect the grant dates for equity awards, and Sangamo does not time the release of material non-public information based on equity award grant dates.
Summary Compensation Table
The following table provides certain summary information for the years ended December 31,
2018, 20172020, 2019 and
2016,2018, when applicable, concerning the compensation earned by (i) our President and Chief Executive Officer,
41
(ii) our former Executive Vice President and Chief Financial Officer, and (iii) our three other most highly compensated executive officers who were serving as executive officers at the end of 2018.2020 and (iv) a former highly compensated executive officer who would be included in the three most highly compensated executive officers in (iii) above, but for the fact that he was not serving as our executive officer at the end of 2020. These individuals are referred to throughout this Proxy Statement as our “named executive officers.” We have omitted certain rows from the table when there is no disclosure of any compensation for the named executive officer for that fiscal year.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name and Principal Position | | Year | | | Salary
($) | | | Bonus
($) | | | | | | Stock
Awards
($) (1) | | | Option
Awards
($) (2) | | | | | | Non-
Equity
Incentive
Plan
Compensation
($) (3) | | | All Other
Compensation
($) (4) | | | | | | Total
($) | |
(a) | | (b) | | | (c) | | | (d) | | | | | | (e) | | | (f) | | | | | | (g) | | | (i) | | | | | | (j) | |
| Alexander D. Macrae, | | | 2018 | | | | 636,480 | | | | - | | | | | | | | 631,575 | | | | 2,410,657 | | | | | | | | 420,077 | | | | 5,639 | | | | | | | | 4,104,428 | |
President and Chief Executive Officer (5) | | | 2017 | | | | 612,000 | | | | - | | | | | | | | - | | | | 795,564 | | | | | | | | 381,888 | | | | 6,404 | | | | | | | | 1,795,856 | |
| | | 2016 | | | | 350,000 | | | | 200,000 | (6) | | | | | | | - | | | | 2,883,090 | | | | | | | | 147,000 | | | | - | | | | | | | | 3,580,090 | |
| | | | | | | | | | | |
| Kathy Y. Yi, | | | 2018 | | | | 404,833 | | | | - | | | | | | | | 145,363 | | | | 554,884 | | | | | | | | 155,861 | | | | 5,639 | | | | | | | | 1,266,580 | |
Executive Vice President and Chief Financial Officer (7) | | | 2017 | | | | 294,358 | | | | - | | | | | | | | - | | | | 574,560 | | | | | | | | 120,377 | | | | 4,710 | | | | | | | | 994,005 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | |
| Heather D. Turner, | | | 2018 | | | | 356,000 | | | | 150,000 | (9) | | | | | | | - | | | | 3,047,160 | | | | | | | | 141,166 | | | | 5,503 | | | | | | | | 3,699,829 | |
Former Senior Vice President and General Counsel (8) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | |
| Stéphane Boissel, | | | 2018 | | | | 120,390 | | | | 100,000 | (12) | | | | | | | - | | | | 2,052,792 | | | | | | | | - | | | | 316,884 | | | | | | | | 2,590,066 | |
Executive Vice President, | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Corporate Strategy (10) (11) | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | |
Edward R Conner, M,D., | | | 2018 | | | | 408,000 | | | | - | | | | | | | | 122,806 | | | | 468,739 | | | | | | | | 154,224 | | | | 1,639 | | | | | | | | 1,555,408 | |
Senior Vice President and Chief | | | 2017 | | | | 400,000 | | | | - | | | | | | | | - | | | | - | | | | | | | | 140,280 | | | | 840 | | | | | | | | 541,120 | |
Medical Officer (13) | | | 2016 | | | | 34,848 | | | | 100,000 | (14) | | | | | | | - | | | | 377,820 | | | | | | | | - | | | | - | | | | | | | | 512,668 | |
| (1) | The amounts in column (e) reflect the aggregate grant date fair value of the RSUs awarded to the named executive officer for the applicable year, calculated in accordance with ASC 718, without taking into account any estimated forfeitures. The assumptions used in the calculation of such grant date fair values are described in Note 4 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the year ended December 31, 2018 filed with the SEC on March 1, 2019.
|
| (2) | The amounts in column (f) represent the aggregate grant date fair value of the stock option grants for each indicated year in accordance with ASC 718 using the Black-Scholes option valuation model and without taking into account any estimated forfeitures. The assumptions used in the calculation of such grant date fair values are described in Note 4 of the Notes to Consolidated Financial Statements included in our Annual Report on Form10-K for the year ended December 31, 2018 filed with the SEC on March 1, 2019.
|
| (3) | The amounts in column (g) reflect the cash bonus awards made to the named executive officer under the cash incentive program under the Incentive Plan for the indicated year.
|
| (4) | The amounts in column (i) include matching payments made to the named executive officer under our 401(k) Plan, a qualified deferred compensation plan under Section 401(k) of the Code ($4,000 for each of Dr. Macrae, Ms. Yi and Ms. Turner), life insurance premiums paid by us on behalf of the named executive officer in the amount of $1,639 for each of Dr. Macrae, Ms. Yi, Dr. Conner and $1,503 for Ms. Turner; and for Mr. Boissel, includes a car allowance in the amount of €1,272 ($1,457), premiums for unemployment insurance contract in the amount of €3,123 ($3,584) that were paid by TxCell subsequent to our acquisition of a controlling interest, as well as a transaction bonus in the amount of €200,210 ($229,360) awarded to Mr. Boissel by TxCell in connection our acquisition of a controlling interest in TxCell, which was paid in November 2018, and Mr. Boissel’s target incentive bonus paid by TxCell of €72,000 ($82,483).
|
| (5) | Dr. Macrae was appointed as our President and Chief Executive Officer on June 1, 2016.
|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name and Principal Position | | Year | | Salary
($) | | Bonus
($) | | | | Stock
Awards
($) (1) | | Option
Awards
($) (2) | | Non-
Equity
Incentive
Plan
Compensation
($) (3) | | All Other
Compensation
($) (4) | | Total
($) |
| (a) | | (b) | | (c) | | (d) | | | | (e) | | (f) | | (g) | | (i) | | (j) |
| Alexander D. Macrae | | 2020 | | 681,797 | | | — | | | | | 1,147,500 | | | 1,488,746 | | | 511,348 | | | 5,639 | | | 3,835,030 | |
| President and | | 2019 | | 661,939 | | | — | | | | | 571,148 | | | 2,274,230 | | | 436,880 | | | 42,358 | | | 3,986,555 | |
| Chief Executive Officer | 2018 | | 636,480 | | | — | | | | | 631,575 | | | 2,410,657 | | | 420,077 | | | 5,639 | | | 4,104,428 | |
| Sung H. Lee | | 2020 | | 438,262 | | | — | | | | | 340,000 | | | 443,316 | | | 215,000 | | (6) | | 5,639 | | | 1,442,217 | |
| Former Executive Vice President | | 2019 | | 72,500 | | | 200,000 | | | (6) | | | 452,813 | | | 1,757,679 | | | 47,415 | | (7) | | 273 | | | 2,530,680 | |
| and Chief Financial Officer (5) | | | | | | | | | | | | | | | | | | |
| Gary H. Loeb | | 2020 | | 406,000 | | | — | | | | | 340,000 | | | 441,110 | | | 200,564 | | | 5,639 | | | 1,393,313 | |
| Executive Vice President, | | 2019 | | 166,667 | | | — | | | | | — | | | 1,788,475 | | | 87,200 | | | 683 | | | 2,043,025 | |
| General Counsel, Corporate Secretary | | | | | | | | | | | | | | | | | | |
| and Chief Compliance Officer (8) | | | | | | | | | | | | | | | | | | |
| D. Mark McClung | | 2020 | | 276,667 | | | 150,000 | | | (10) | | | 916,000 | | | 1,187,220 | | | 136,245 | | | 5,093 | | | 2,671,225 | |
| Executive Vice President, | | | | | | | | | | | | | | | | | | |
| Chief Business Officer (9) | | | | | | | | | | | | | | | | | | |
| R. Andrew Ramelmeier | | 2020 | | 416,000 | | | 50,000 | | | (12) | | | 374,000 | | | 485,221 | | | 203,840 | | | 5,639 | | | 1,534,700 | |
| Executive Vice President, | | 2019 | | 382,965 | | | — | | | | | 112,875 | | | 449,453 | | | 153,608 | | | 5,639 | | | 1,104,540 | |
| Technical Operations (11) | | | | | | | | | | | | | | | | | | |
| Stéphane Boissel | | 2020 | | 318,283 | | | — | | | | | 374,000 | | | 485,221 | | | — | | | 256,472 | | | 1,433,976 | |
| Former Executive Vice President, | | 2019 | | 483,600 | | | — | | | | | 158,025 | | | 629,234 | | | 212,784 | | | 1,639 | | | 1,485,282 | |
| Corporate Strategy (13) (14) | | 2018 | | 120,390 | | | 100,000 | | | (15) | | | — | | | 2,052,792 | | | — | | | 316,884 | | | 2,590,066 | |
___________________
(1)The amounts in column (e) reflect the aggregate grant date fair value of the RSUs awarded to the named executive officer for the applicable year, calculated in accordance with ASC 718, without taking into account any estimated forfeitures. The grant date fair value of the RSUs is measured based on the closing price of the underlying common stock on the date of grant.
(2)The amounts in column (f) represent the aggregate grant date fair value of the stock option grants for each indicated year in accordance with ASC 718 using the Black-Scholes option valuation model and without taking into account any estimated forfeitures. The assumptions used in the calculation of such grant date fair values are described in Note 9 of the Notes to Consolidated Financial Statements included in the 2020 Form 10-K.
(3)The amounts in column (g) reflect the cash bonus awards made to the named executive officer under the Cash Incentive Compensation Program under the Incentive Plan for the indicated year.
(4)The amounts in column (i) for 2020 include matching payments of $4,000 made to each named executive officer under our 401(k) Plan, a qualified deferred compensation plan under Section 401(k) of the Code; life insurance premiums paid by us on behalf of the named executive officer in the amount of $1,639 for each of Dr. Macrae, Mr. Lee, Mr. Loeb and Dr. Ramelmeier, $1,093 for Mr. McClung and $956 for Mr. Boissel; a severance payout of $230,516 for Mr. Boissel; and a $25,000 payment to Mr. Boissel for consulting services rendered to the Company subsequent to his termination.
(5)Mr. Lee was appointed as our Executive Vice President and Chief Financial Officer effective October 31, 2019 and resigned effective February 1, 2021.
(6)Consists of his annual incentive cash compensation under the Incentive Plan paid to Mr. Lee pursuant to the terms of his consulting agreement under which he agreed to provide consulting services to us through February 26, 2021 following his resignation effective February 1, 2021.
(7)Consists of a sign-on bonus that was subject to repayment in the event Mr. Lee’s employment was terminated for cause or by him without good reason within one year of his appointment.
(8)Mr. Loeb was appointed as our Executive Vice President and General Counsel effective July 30, 2019 and as our Corporate Secretary and Chief Compliance Officer effective September 16, 2019.
| (6) | Consists of asign-onTable of Contents bonus that was subject to repayment in the event Dr. Macrae’s employment was terminated for cause or by him without good reason within one year of his appointment.
|
| (7) | Ms. Yi was appointed as our Senior Vice President and Chief Financial Officer on February 28, 2017.
|
| (8) | Ms. Turner was appointed as our Senior Vice President and General Counsel and Secretary on February 12, 2018 and resigned effective on March 15, 2019.
|
| (9) | Consists of asign-on bonus that was subject to repayment in the event Ms. Turner’s employment was terminated for cause or by her without good reason within one year of her appointment.
|
| (10) | Mr. Boissel joined our company on October 2, 2018 in connection with the TxCell acquisition and was employed on a part-time basis through December 31, 2018 by us, and received a €25,000 monthly salary from TxCell during such period, in addition to his car allowance and insurance premiums noted in footnote (4) above.
|
| (11) | Certain amounts paid to Mr. Boissel were paid by TxCell in Euro, and converted to U.S. dollars at an exchange rate of €1.00 = $1.1456, the noon buying rate as reported by the Federal Reserve Bank of New York on December 31, 2018.
|
| (12) | Consists of asign-on bonus that is subject to repayment in the event Mr. Boissel’s employment is terminated for cause or by him without good reason within one year of his appointment.
|
| (13) | Dr. Conner commenced employment on November 30, 2016.
|
| (14) | Consists of asign-on bonus that was subject to repayment in the event Dr. Conner’s employment was terminated for cause or by him without good reason within one year of his appointment.
|
43
(9)Mr. McClung was appointed as our Executive Vice President, Chief Business Officer effective May 1, 2020.
(10)Consists of a sign-on bonus that is subject to repayment in the event Mr. McClung’s employment is terminated for cause or by him without good reason within one year of his appointment.
(11)Dr. Ramelmeier was promoted to the position of Executive Vice President, Technical Operations effective September 16, 2019. Dr. Ramelmeier was hired on January 1, 2018 as our Senior Vice President, Technical Operations and did not serve as an executive officer of Sangamo during any portion of 2018.
(12)In December 2019, the Compensation Committee approved a one-time special retention bonus of $100,000 to be paid to Dr. Ramelmeier in four equal payments of $25,000 in July 2020, December 2020, July 2021 and December 2021.
(13)Mr. Boissel joined our company on October 2, 2018 in connection with the acquisition of Sangamo France, as our Executive Vice President, Corporate Strategy and resigned effective July 31, 2020.
(14)In 2018, certain amounts paid to Mr. Boissel were paid by Sangamo France in Euro and converted to U.S. dollars at an exchange rate of €1.00 = $1.1456, the noon buying rate as reported by the Federal Reserve Bank of New York on December 31, 2018.
(15)Consists of a sign-on bonus that was subject to repayment in the event Mr. Boissel’s employment was terminated for cause or by him without good reason within one year of his appointment.
Grants of Plan-Based Awards
The following table shows for the fiscal year ended December 31,
2018,2020, certain information regarding grants of plan-based awards to our named executive officers.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | Estimated Possible Payouts Under Non-Equity
Incentive Plan Awards | | | All
Other
Stock
Awards:
Number
of
Shares
of Stock
or Units
(#) (2) | | | All Other
Option
Awards:
Number of
Securities
Underlying
Options
(#) (3) | | | Exercise or
Base
Price of
Option or
Stock
Awards
($/Sh) | | | Grant
Date
Fair Value
of Stock
and
Option
Awards
($) (4) | |
Name | | Award Type | | Grant
Date | | | Approval Date | | | Threshold
($) (1) | | | Target
($) (1) | | | Maximum
($) (1) | |
(a) | | | | (b) | | | | | | (c) | | | (d) | | | (e) | | | (j) | | | (k) | | | (l) | | | (m) | |
Alexander D. Macrae | | Annual Cash | | | | | | | | | | | - | | | | 381,888 | | | | 763,776 | | | | - | | | | - | | | | - | | | | - | |
| | Annual Grant | | | 1/24/2018 | | | | 01/24/2018 | | | | - | | | | - | | | | - | | | | 31,500 | | | | 189,000 | | | | 20.05 | | | | 3,042,232 | |
Kathy Y. Yi | | Annual Cash | | | | | | | | | | | - | | | | 141,692 | | | | 283,384 | | | | - | | | | - | | | | - | | | | | |
| | Annual Grant | | | 1/24/2018 | | | | 01/24/2018 | | | | | | | | | | | | | | | | 7,250 | | | | 43,500 | | | | 20.05 | | | | 700,247 | |
Heather D. Turner | | Annual Cash | | | | | | | | | | | - | | | | 128,333 | | | | 256,666 | | | | - | | | | - | | | | - | | | | - | |
| | New Hire Grant | | | 2/28/2018 | | | | 1/24/2018 | | | | - | | | | | | | | | | | | - | | | | 200,000 | | | | 23.95 | | | | 3,047,160 | |
Stéphane Boissel | | New Hire Grant | | | 11/23/2018 | | | | 09/24/2018 | | | | - | | | | - | | | | - | | | | - | | | | 215,000 | | | | 11.08 | | | | 1,556,665 | |
| | New Hire Grant (5) | | | 11/23/2018 | | | | 09/24/2018 | | | | - | | | | - | | | | - | | | | - | | | | 68,523 | | | | 11.08 | | | | 496,127 | |
Edward R. Conner, M.D. | | Annual Cash | | | | | | | | | | | - | | | | 142,800 | | | | 285,600 | | | | - | | | �� | - | | | | - | | | | - | |
| | Annual Grant | | | 1/24/2018 | | | | 01/24/2018 | | | | - | | | | | | | | | | | | 6,125 | | | | 36,750 | | | | 20.05 | | | | 591,545 | |
(1) | The dollar amounts represent the threshold, target and maximum amounts of each named executive officer’s potential annual cash bonus award for the year ended December 31, 2018 pursuant to the 2018 Cash Incentive Compensation Program under the Incentive Plan, and in Ms. Turner’s case, also reflectpro-ration as she commenced employment in February 2018. The amount shown as target reflects the target payment level if Sangamo and each individual had achieved 100% of the corporate and individual performance goals established pursuant to the Incentive Plan. Each of Ms. Yi’s, Dr. Conner’s, and Ms. Turner’s target bonuses were allocated between corporate and individual performance (80% to 20%, respectively), and Dr. Macrae’s target bonus was allocated 100% to corporate performance. The Compensation Committee determines the level of attainment of the corporate performance objectives following the end of the performance period. The amount shown as maximum reflects the payment level pursuant to the 2018 Cash Incentive Program if Sangamo had achieved a 200% corporate performance percentage and each individual (other than Dr. Macrae) had achieved a 200% individual performance percentage, which were the maximum percentages allowed for the corporate and individual performance percentages. Actual payouts differed based on the actual performance objectives achieved. The actual cash bonus award earned for the year ended December 31, 2018 pursuant to the 2018 Cash Incentive Program for each named executive officer is set forth in the Summary Compensation Table above. As such, the amounts set forth in these columns do not represent additional compensation earned by the named executive officers for the year ended December 31, 2018. For more information regarding the 2018 Cash Incentive Compensation Program, see “—Compensation Discussion and Analysis—2018 Compensation Decisions—2018 Cash Incentive Compensation” above and “—Employment Agreements and Compensation Arrangements—Annual Cash Bonus Awards” below. Mr. Boissel joined the Company in October 2018 on a part-time basis through December 31, 2018 and was not eligible to participate in this program during 2018.
|
44
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | |
Estimated Future Payouts Under Non-Equity Incentive Plan Awards | | All
Other
Stock
Awards:
Number
of
Shares
of Stock
or Units
(#) (2) | | All Other
Option
Awards:
Number of
Securities
Underlying
Options
(#) (3) | | Exercise or
Base
Price of
Option or
Stock
Awards
($/Sh) | | Grant
Date
Fair Value
of Stock
and
Option
Awards
($) (4) |
| Name | | Award Type | | Grant
Date | | Approval
Date | | Target
($) (1) | | Maximum
($) (1) | |
| (a) | | | | (b) | | | | (d) | | (e) | | (j) | | (k) | | (l) | | (m) |
| Alexander D. Macrae | | Annual Cash | | | | | | 409,078 | | | 818,156 | | | — | | | — | | | — | | | — | |
| | Annual RSU Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | 168,750 | | | — | | | — | | | 1,147,500 | |
| | Annual Option Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | — | | | 337,500 | | | 6.80 | | 1,488,746 | |
| Sung H. Lee | | Annual Cash | | | | | | 175,305 | | | 350,610 | | | — | | | — | | | — | | | — | |
| | Annual RSU Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | 50,000 | | | — | | | — | | | 340,000 | |
| | Annual Option Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | — | | | 100,500 | | | 6.80 | | 443,316 | |
| Gary H. Loeb | | Annual Cash | | | | | | 162,400 | | | 324,800 | | | — | | | — | | | — | | | — | |
| | Annual RSU Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | 50,000 | | | — | | | — | | | 340,000 | |
| | Annual Option Grant | | 02/25/2020 | | 12/19/2019 | | — | | | — | | | — | | | 100,000 | | | 6.80 | | 441,110 | |
| D. Mark McClung | | Annual Cash | | | | | | 110,667 | | | 221,334 | | | — | | | — | | | — | | | — | |
| | New Hire RSU Grant | | 06/25/2020 | | 04/10/2020 | | — | | | — | | | 100,000 | | | — | | | — | | | 916,000 | |
| | New Hire Option Grant | | 06/25/2020 | | 04/10/2020 | | — | | | — | | | — | | | 200,000 | | | 9.16 | | 1,187,220 | |
| R. Andrew Ramelmeier | | Annual Cash | | | | | | 166,400 | | | 332,800 | | | — | | | — | | | — | | | — | |
| | Annual RSU Grant | | 02/25/2020 | | 01/30/2020 | | — | | | — | | | 55,000 | | | — | | | — | | | 374,000 | |
| | Annual Option Grant | | 02/25/2020 | | 01/30/2020 | | — | | | — | | | — | | | 110,000 | | | 6.80 | | 485,221 | |
| Stéphane Boissel | | Annual Cash | | | | | | 201,178 | | | 402,356 | | | — | | | — | | | — | | | — | |
| | Annual RSU Grant | | 02/25/2020 | | 01/30/2020 | | — | | | — | | | 55,000 | | | — | | | — | | | 374,000 | |
| | Annual Option Grant | | 02/25/2020 | | 01/30/2020 | | — | | | — | | | — | | | 110,000 | | | 6.80 | | 485,221 | |
___________________
(1)The dollar amounts represent the target and maximum amounts of each named executive officer’s potential annual cash bonus award for the year ended December 31, 2020 pursuant to the 2020 Cash Incentive Compensation Program under the Incentive Plan. In Mr. McClung’s case, the amounts reflect proration as he commenced employment in May 2020. The amount shown as target reflects the target payment level if Sangamo and each individual had achieved 100% of the corporate and individual performance goals established pursuant to the Incentive Plan. Each of Mr. Lee’s, Mr. Loeb’s, Mr. McClung’s, Dr. Ramelmeier’s and Mr. Boissel’s target bonuses were allocated between corporate and individual performance (90% to 10%, respectively), and Dr. Macrae’s target bonus was allocated 100% to corporate performance. The Compensation Committee determines the level of attainment of the corporate performance objectives following the end of the performance period. The amount shown as maximum reflects the payment level pursuant to the 2020 Cash Incentive Compensation Program if Sangamo had achieved a 200% corporate performance percentage and each individual (other than Dr. Macrae) had achieved a 200% individual performance percentage, which were the maximum percentages allowed for the corporate and individual performance percentages. Actual payouts differed based on the actual performance objectives achieved. The actual cash bonus award earned for the year ended December 31, 2020 pursuant to the 2020 Cash Incentive Compensation Program for each named executive officer is set forth in the Summary Compensation Table above. As such, the amounts set forth in these columns do not represent additional compensation earned by the named executive officers for the year ended December 31, 2020. For more information regarding the 2020 Cash Incentive
(2) | The reported RSU award was granted under our Amended and Restated 2013 Stock Incentive Plan, or the 2013 Plan, and will vest and become exercisable in accordance with the following schedule: 1/3rd of the shares vest in three equal annual installments over the three-year period measured from the grant date, provided the grantee continues to provide services through each applicable vesting date.
|
(3) | With the exception of Mr. Boissel, the reported option was granted under the 2013 Plan and will vest and become exercisable in accordance with the following schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the36-Table of Contents month period measured from the first anniversary of the option grant date, provided the optionee continues to provide services through each applicable vesting date. The reported option for Mr. Boissel was granted under the 2018 Plan and will vest in the same manner as the aforementioned options granted under the 2013 Plan.
|
(4) | Represents the grant date fair value of such stock option and RSU as determined in accordance with ASC 718. The assumptions used in the calculation of the grant date fair values of each grant are included in Note 4 of the Notes to Consolidated Financial Statements in our Annual Report on Form10-K for the year ended December 31, 2018 filed with the SEC on March 1, 2019.
|
(5) | Represents options granted to Mr. Boissel as an employee of TxCell, in connection with the Company’s acquisition of TxCell.
|
45
Compensation Program, see “—Compensation Discussion and Analysis—2020 Compensation Decisions—2020 Cash Incentive Compensation” above and “—Employment Agreements and Compensation Arrangements—Annual Cash Bonus Awards” below.
(2)The reported RSU award was granted under the 2018 Plan and will vest and become exercisable in accordance with the following schedule: 1/3rd of the shares vest in three equal annual installments over the three-year period measured from the grant date, subject to the grantee’s continuous service through each applicable vesting date.
(3)The reported option was granted under the 2018 Plan and will vest and become exercisable in accordance with the following schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the option grant date, subject to the optionee’s continuous service through each applicable vesting date.
(4)Represents the grant date fair value of such stock option and RSU as determined in accordance with ASC 718. The grant date fair value of the RSUs is measured based on the closing price of the underlying common stock on the date of grant. The assumptions used in the calculation of the grant date fair values of stock options using the Black-Scholes option valuation model are included in Note 9 of the Notes to Consolidated Financial Statements in the 2020 Form 10-K.
Employment Agreements and Compensation Arrangements
Each of our named executive officers has entered into a written employment agreement with our company. For a description of these employment agreements, please see the section of this Proxy Statement under the heading “—Employment Contracts and
Separation and Change in Control Arrangements” below.
In March 2018, the Compensation Committee of the Board adopted the Incentive Plan, which amended and restated the prior plan adopted in
2012, and2012. The Incentive Plan is a performance-based cash incentive program for
our eligible employees, including our executive officers. Under the Incentive Plan, unless the Compensation Committee determines otherwise, each performance period
shall beis coincident with the
calendar year, commencing with the 2018 calendar year. The Compensation Committee establishes the specific corporate objectives and/or individual objectives that must be attained in order for Incentive Plan participants to receive a bonus for that performance period. For each performance and individual objective, the Compensation Committee may set threshold and target levels of attainment. The Compensation Committee will then establish for each participant dollar levels for the bonus to which he or she may become entitled for that performance period based on the level at which the corporate objectives and/or individual objectives are actually attained.
Performance objectives established under the Incentive Plan are based on one or more of the following criteria: (i) revenue, organic revenue, net sales, or
new-product revenue,
or net sales, (ii) achievement of specified milestones in the discovery and development of the Company’s technology or of one or more of the Company’s products, (iii) achievement of specified milestones in the commercialization of one or more of the Company’s products, (iv) achievement of specified milestones in the manufacturing of one or more of the Company’s products, (v) expense targets, (vi) share price, (vii) total stockholder return, (viii) earnings per share, (ix) operating margin, (x) gross margin, (xi) return measures (including, but not limited to, return on assets, capital, equity, or sales), (xii) productivity ratios, (xiii) operating income, (xiv) net operating profit, (xv) net earnings or net income (before or after taxes), (xvi) cash flow (including, but not limited to, operating cash flow, free cash flow and cash flow return on capital), (xvii) earnings before or after interest, taxes, depreciation, amortization and/or stock-based compensation expense, (xviii) economic value added, (xix) market share, (xx) working capital targets, (xxi) achievement of specified milestones relating to corporate partnerships, collaborations, license transactions, distribution arrangements, mergers, acquisitions, dispositions or similar business transactions, (xxii) employee retention and recruiting and human resources management, and (xxiii) other corporate performance criteria approved by the Compensation Committee.
Individual performance objectives established under the Incentive Plan are based on one or more of the following criteria: (i) the participant’s contribution toward the achievement of a specific corporate objective, (ii) the contribution of the business unit or division supervised by the participant toward the achievement of a specific corporate objective, and (iii) the participant’s development of professional skills.
A participant will not become entitled to a
cash bonus under the Incentive Plan for a particular performance period unless the participant is employed
on or before October
31st1st of the applicable performance period. Following the close of the applicable performance period, the Compensation Committee will determine the actual bonus amount for each participant based on the actual levels at which the actual performance objectives are attained, and bonuses will be paid promptly following the end of the applicable performance period.
The 2020 criteria for the Incentive Plan required continuous service through the date of the Compensation Committee’s determination of the level of attainment of the corporate performance objectives. Mr. Lee’s cash bonus under the Incentive Plan was determined while Mr. Lee was an eligible participant under such plan and paid to Mr. Lee pursuant to the terms of his consulting agreement in which he provided consulting services to us through February 26, 2021 following his February 1, 2021 termination of employment.
In the event any payment to which a participant becomes entitled under the Incentive Plan would otherwise constitute a parachute payment under Section 280G of the Code, then that payment will be subject to reduction to the extent necessary to assure that such payment will be limited to the greater of (i) the dollar amount that can be paid to the participant without triggering a parachute payment under Code Section 280G or (ii) the dollar amount of that payment which provides the participant with the greatest
after-tax amount after taking into account any excise tax the participant may incur under Code Section 4999 with respect to such payment and any other benefits or payments to which the participant may be entitled in connection with any change in control of the Company or the subsequent termination of the participant’s employment.
46
We adopted a 20182020 Cash Incentive Compensation Program under the Incentive Plan that provides for annual bonus awards to reward executive officers based on our achievement of specific corporate goals and their achievement of individual performance goals. For more information regarding the 20182020 Cash Incentive Compensation Program under the Incentive Plan, please see the section of this Proxy Statement under the heading “—Compensation Discussion and Analysis—20182020 Compensation Decisions—20182020 Cash Incentive Compensation” and footnote (1) to the Grants of Plan-Based Awards table above.
Discretionary options that we granted in
20182020 under the
20132018 Plan vest according to the following schedule: 25% of the option shares will vest and become exercisable on the
one yearone-year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the
36-month period measured from the first anniversary of the option grant date,
providedsubject to the executive
officer continues to provide servicesofficer’s continuous service through each applicable vesting date. Each option will vest on an accelerated basis in connection with certain terminations and changes, as described under the heading “—Employment Contracts and
Change in Control Agreements” below. Each option has an expiration date at the end of the10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service. Options granted under the 2013 Plan have an exercise price equal to the fair market value on the date of grant (generally the closing price of our common stock on the grant date on the Nasdaq Global Select Market), and the 2013 Plan permits the exercise price of stock options to be paid by cash, check, other shares of our common stock (with some restrictions), brokerassisted same-day sales, and cashless “net exercise” arrangements. Discretionary RSUs that we granted in 2018 under the 2013 Plan vest in three successive equal annual installments over the three-year period measured from the grant date, provided the executive officer continues to provide services through each applicable vesting date. For a discussion of the effect on outstanding equity awards under the 2013 Plan in connection with certain terminations and changes in control of the Company, including accelerated vesting, see the discussion under the heading “—Employment Contracts and Change in Control Arrangements” below.Discretionary options that we granted in 2018 under the 2018 Plan vest according to the following schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the36-month period measured from the first anniversary of the option grant date, provided the executive officer continues to provide services through each applicable vesting date. Each option will vest on an accelerated basis in connection with certain terminations and changes, as described under the heading “—Employment ContractsSeparation and Change in Control Agreements” below. Each option has an expiration date at the end of the10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service. Options granted under the 2018 Plan have an exercise price equal to the fair market value on the date of grant (generally the closing price of our common stock on the grant date on the Nasdaq Global Select Market), and the 2018 Plan permits the exercise price of stock options to be paid by cash, check, other shares of our common stock (with some restrictions), brokerassisted same-day sales, and cashless “net exercise” arrangements. AlthoughDiscretionary RSUs that we did not grant any discretionary RSUsgranted in 20182020 under the 2018 Plan grants of discretionary RSUs in February 2019 to executive officers under such plan vest in three successive equal annual installments over the three-year period measured from the grant date, providedsubject to the executive officer continues to provide servicesofficer’s continuous service through each applicable vesting date. For a discussion of the effect on outstanding equity awards under the 2018 Plan in connection with certain terminations and changes in control of the Company, including accelerated vesting, see the discussion under the heading “—Employment Contracts and Separation and Change in Control Arrangements” below.
Other Compensatory Arrangements
401(k) Plan. Our executive officers are eligible to participate in the 401(k) Plan. The 401(k) Plan is intended to qualify as atax-qualified plan under Section 401 of the Code. The 401(k) Plan provides that each participant may contribute a portion of his or herpre-tax compensation, up to a statutory annual limit. The 401(k) Plan also permits us to make discretionary contributions and matching contributions, subject to established limits. In 2018,2020, we made a matching contribution, which was subject to an annual limit of $4,000 per employee.47
Other Benefits. Our executive officers are eligible to participate in all of our benefit plans, such as our medical, dental, vision, short-term disability, long-term disability and group life insurance plans and our employee stock purchase plans (including the Purchase Plan,2020 ESPP), in each case generally on the same basis as other employees. We also have a Section 125 flexible benefits healthcare plan and a flexible benefits childcare plan under which employees can set asidepre-tax funds to pay for qualified healthcare expenses and qualified childcare expenses not reimbursed by insurance, respectively. We do not currently offer pension or other retirement benefits in the United States but do offer pension or other retirement benefits in certain other countries.
Outstanding Equity Awards at Fiscal
Year-EndThe following table provides certain summary information concerning outstanding equity awards held by the named executive officers as of December 31,
2018. | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Option Awards | | | Stock Awards | |
Name | | Grant Date | | | Number of
Securities
Underlying
Unexercised
Options (#)
Exercisable
(1) | | | Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable
(1) | | | Option
Exercise
Price
($) | | | Option
Expiration
Date | | | Number
of
Shares
or
Units of
Stock
that
Have
Not
Vested
(#) | | | Market
Value of
Shares or
Units of
Stock
That
Have Not
Vested
($) (3) | |
(a) | | | | | (b) | | | (c) | | | (d) | | | (e) | | | (f) (2) | | | (g) | |
Alexander D. Macrae | | | 01/24/2018 | | | | - | | | | 189,000 | | | | 20.05 | | | | 01/23/2028 | | | | 31,500 | | | | 361,620 | |
| | | 01/26/2017 | | | | 172,500 | | | | 187,500 | | | | 3.50 | | | | 01/25/2027 | | | | - | | | | | |
| | | 06/03/2016 | | | | 437,500 | | | | 262,500 | | | | 7.07 | | | | 06/02/2026 | | | | - | | | | - | |
Kathy Y. Yi | | | 01/24/2018 | | | | - | | | | 43,500 | | | | 20.05 | | | | 01/23/2028 | | | | 7,250 | | | | 83,230 | |
| | | 02/28/2017 | | | | 41,666 | | | | 108,334 | | | | 4.55 | | | | 02/27/2027 | | | | - | | | | - | |
| Heather D. Turner (4) | | | 2/28/2018 | | | | - | | | | 200,000 | | | | 23.95 | | | | 02/27/2028 | | | | - | | | | - | |
Stéphane Boissel | | | 11/23/2018 | | | | - | | | | 283,523 | | | | 11.08 | | | | 11/23/2028 | | | | - | | | | - | |
| Edward R. Conner, M.D. | | | 01/24/2018 | | | | - | | | | 36,750 | | | | 20.05 | | | | 01/23/2028 | | | | 6,125 | | | | 70,315 | |
| | | 11/30/2016 | | | | 39,166 | | | | 95,834 | | | | 3.20 | | | | 11/29/2026 | | | | - | | | | - | |
(1) | Except as otherwise provided in the footnotes below, each option was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the36- month period measured from the first anniversary of the option grant date, provided the executive officer continues to provide services through each applicable vesting date. Each option will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Change in Control Agreements.” Each option has an expiration date at the end of the10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service.
|
(2) | Represents an RSU award subject to vesting in three successive equal annual installments over the three-year period measured from the grant date, provided the executive officer continues to provide services to the Company through each applicable vesting date. The award will vest on an accelerated basis in connection with certain terminations and changes in control of the Company, as described under the heading “—Employment Contracts and Change in Control Arrangements.”
|
(3) | Based on the $11.48 closing price of our common stock on December 31, 2018, the last trading day of fiscal 2018.
|
(4) | Ms. Turner’s option was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of employment start date (February 12, 2019) and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the36- month period measured from the first anniversary of the employment start date, provided the executive officer continues to provide services through each applicable vesting date. The option will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Change in Control Agreements.” The option has an expiration date at the end of the10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service.
|
48
2020. | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | Option Awards | | Stock Awards |
| Name | | Grant Date | | Number of
Securities
Underlying
Unexercised
Options (#)
Exercisable
(1) | | Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable
(1) | | Option
Exercise
Price
($) | | Option
Expiration
Date | | Equity Incentive Plan Awards:
Number of
Unearned Shares,
Units or Other Rights
That
Have Not
Vested
(#) (2) | | Equity Incentive Plan Awards:
Market or Payout
Value of
Unearned Shares,
Units or Other Rights
That
Have Not
Vested
($) (3) |
| (a) | | | | (b) | | (c) | | (d) | | (e) | | (f) | | (g) |
| Alexander D. Macrae | | 02/25/2020 | | — | | | 337,500 | | | 6.80 | | | 02/24/2030 | | — | | | — | |
| | 02/25/2019 | | 179,937 | | | 205,563 | | | 9.03 | | | 02/24/2029 | | — | | | — | |
| | 01/24/2018 | | 137,812 | | | 51,188 | | | 20.05 | | | 01/23/2028 | | — | | | — | |
| | 01/26/2017 | | 352,500 | | | 7,500 | | | 3.50 | | | 01/25/2027 | | — | | | — | |
| | 06/03/2016 | | 700,000 | | | — | | | 7.07 | | | 06/02/2026 | | — | | | — | |
| | 02/25/2020 | | — | | | — | | | — | | | | | 168,750 | | | 2,634,188 | |
| | 02/25/2019 | | — | | | — | | | — | | | | | 42,168 | | | 658,242 | |
| | 01/24/2018 | | — | | | — | | | — | | | | | 10,500 | | | 163,905 | |
| Sung H. Lee (4) | | 02/25/2020 | | — | | | 100,500 | | | 6.80 | | | 02/24/2030 | | — | | | — | |
| | 11/25/2019 | | 70,958 | | | 191,042 | | | 10.35 | | | 11/24/2029 | | — | | | — | |
| | 02/25/2020 | | — | | | — | | | — | | | | | 50,000 | | | 780,500 | |
| | 11/25/2019 | | — | | | — | | | — | | | | | 29,168 | | | 455,312 | |
| Gary H. Loeb (5) | | 02/25/2020 | | — | | | 100,000 | | | 6.80 | | | 02/24/2030 | | — | | | — | |
| | 08/23/2019 | | 83,333 | | | 166,667 | | | 11.02 | | | 08/22/2029 | | — | | | — | |
| | 02/25/2020 | | — | | | — | | | — | | | | | 50,000 | | | 780,500 | |
| D. Mark McClung (6) | | 06/25/2020 | | — | | | 200,000 | | | 9.16 | | | 06/24/2030 | | — | | | — | |
| | 06/25/2020 | | — | | | — | | | — | | | | | 100,000 | | | 1,561,000 | |
| R. Andrew Ramelmeier | | 02/25/2020 | | — | | | 110,000 | | | 6.80 | | | 02/24/2030 | | — | | | — | |
| | 02/25/2019 | | 34,375 | | | 40,625 | | | 9.03 | | | 02/24/2029 | | — | | | — | |
| | 01/31/2018 | | 87,500 | | | 32,500 | | | 20.85 | | | 01/30/2028 | | — | | | — | |
| | 02/25/2020 | | — | | | — | | | — | | | | | 55,000 | | | 858,550 | |
| | 02/25/2019 | | — | | | — | | | — | | | | | 8,333 | | | 130,078 | |
___________________
(1)Except as otherwise provided in the footnotes below, each option was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of the option grant date and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the option grant date, subject to the executive officer’s continuous service through each applicable vesting date. Each option will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Separation and Change in Control Agreements.” Each option has an expiration date at the end of the 10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service.
(2)Represents a RSU award subject to vesting in three successive equal annual installments over the three-year period measured from the grant date, subject to the executive officer’s continuous service through each applicable vesting date. The award will vest on an accelerated basis in connection with certain terminations and changes in control of the Company, as described under the heading “—Employment Contracts and Separation and Change in Control Arrangements.”
(3)Based on the $15.61 closing price of our common stock on December 31, 2020, the last trading day of fiscal 2020.
(4)Mr. Lee’s option granted November 25, 2019 was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of employment start date (October 31, 2020) and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the employment start date, subject to the executive officer’s continuous service through each applicable vesting date. The RSU award vests in a series of three successive equal installments upon completion of each year of service measured from the grant date, subject to Mr. Lee’s continuous service through each applicable vesting date. The option and RSU will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Separation and Change in Control Agreements.” The option has an expiration date at the end of the 10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service. Mr. Lee resigned effective February 1, 2021, accordingly, his options and RSU awards ceased vesting on that date and expired on May 27, 2021.
(5)Mr. Loeb’s option granted August 23, 2019 was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one year anniversary of employment start date (July 30, 2020) and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the employment start date, subject to the executive officer’s continuous service through each applicable vesting date. The option will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Separation and Change in Control Agreements.” The option has an expiration date at the end of the 10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service.
(6)Mr. McClung’s option granted June 25, 2020 was subject to the following vesting schedule: 25% of the option shares will vest and become exercisable on the one-year anniversary of employment start date (May 1, 2021) and the remaining option shares will vest and become exercisable in 36 equal monthly installments over the 36-month period measured from the first anniversary of the employment start date, subject to Mr. McClung’s continuous service through each applicable vesting date. The RSU award vests in a series of three successive equal installments upon completion of each year of service measured from the grant date, subject to Mr. McClung’s continuous service through each applicable vesting date. The option and RSU will vest on an accelerated basis in connection with certain terminations and changes in control, as described under the heading “—Employment Contracts and Separation and Change in Control Agreements.” The option has an expiration date at the end of the 10-year period measured from the grant date, unless terminated earlier following the optionee’s termination of service.
Option Exercises and Stock Vested
The following table sets forth the number of shares of our common stock acquired and the value realized upon the vesting of RSU awards and on each exercise of stock options for each of the named executive officers during the year ended December 31,
2018: | | | | | | | | | | | | | | | | |
Name | | Option Awards | | | Stock Awards | |
| | Number of
Shares
Acquired
on Exercise (#) | | | Value Realized
on Exercise ($) (1) | | | Number of
Shares
Acquired on
Vesting
(#) | | | Value Realized
on
Vesting ($) | |
(a) | | (b) | | | (c) | | | (d) | | | (e) | |
Alexander D. Macrae | | | - | | | | - | | | | - | | | | - | |
Kathy Y. Yi | | | 50,000 | | | | 536,990 | | | | - | | | | - | |
Heather D. Turner | | | - | | | | - | | | | - | | | | - | |
Stéphane Boissel | | | - | | | | - | | | | - | | | | - | |
Edward R. Conner, M.D. | | | 60,000 | | | | 765,026 | | | | - | | | | - | |
(1) | Value realized is determined by multiplying (i) the amount by which the market price of the common stock on the date of exercise exceeded the exercise price by (ii) the number of shares for which the options were exercised.
|
2020:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | | Option Awards | | Stock Awards |
Number of
Shares
Acquired
on Exercise (#) | | Value Realized
on Exercise ($) (1) | | Number of
Shares
Acquired on
Vesting (#) | | Value Realized
on
Vesting ($) (2) |
| (a) | | (b) | | (c) | | (d) | | (e) |
| Alexander D. Macrae | | — | | | — | | | 31,582 | | | 222,108 | |
| Sung H. Lee | | — | | | — | | | 14,582 | | | 142,466 | |
| Gary H. Loeb | | — | | | — | | | — | | | — | |
| D. Mark McClung | | — | | | — | | | — | | | — | |
| R. Andrew Ramelmeier | | — | | | — | | | 4,167 | | | 28,336 | |
| Stéphane Boissel | | 155,321 | | | 258,977 | | | 5,822 | | | 39,664 | |
___________________
(1)Value realized is determined by multiplying (i) the amount by which the market price of the common stock on the date of exercise exceeded the exercise price by (ii) the number of shares for which the options were exercised.
(2)Value realized is determined by multiplying (i) the market price of the common stock on the date of vesting by (ii) the number of shares acquired on vesting.
We do not sponsor a
tax-qualified defined benefit retirement plan or a supplemental executive retirement plan.
Nonqualified Deferred Compensation
We do not sponsor a nonqualified deferred compensation plan.
Risk Assessment of Compensation Policies and Practices
Our compensation programs throughout the organization are designed to maintain an appropriate balance between long-term and near-term incentives by utilizing a combination of compensation components, including base salary, annual cash incentive awards, and long-term equity awards. Although not all employees in the organization may have compensation comprising all three components, the compensation programs are generally structured so that any near-term cash incentives are not likely to constitute the predominant element of an employee’s total compensation. The Compensation Committee annually reviews our compensation policies and practices to assess whether they encourage employees to take inappropriate risks. After reviewing each of our compensation plans, and the checks and balances built into, and oversight of, each plan, in April 2019,February 2021, the Compensation Committee determined that any risks arising from our compensation policies and practices for our employees are not reasonably likely to have a material adverse effect on us as a whole. In addition, the Compensation Committee believes that the mix and design of the elements of executive compensation do not encourage management to assume excessive risks, and significant compensation decisions, as well as decisions concerning the compensation of our
executive officers, include subjective considerations by the compensation committee or the board of directors, which restrain the influence of formulae or objective factors on excessive risk taking. Finally, the mix of short-term compensation (in the form of salary and annual bonus, if any) and long-term compensation (in the form of stock options and RSUs) also prevents undue focus on short-term results and helps align the interests of our executive officers with the interests of our stockholders.
49
Employment Contracts
and Separation and Change in Control Arrangements
Chief Executive Officer Employment Agreement
In May 2016 we entered into an employment agreement with Dr. Macrae that took effect June 1, 2016, which sets forth the terms and conditions of his employment as President and Chief Executive Officer. Pursuant to his employment agreement, Dr. Macrae will receive a base salary
(initially $600,000 per year) and an annual cash bonus based upon our company’s achievement of specified objectives under our Incentive Plan with a target cash bonus based on a percentage of his base
salary (initially 60%).salary. Dr. Macrae’s base salary and target bonus percentage are subject to annual review by the Compensation Committee and adjustment from time to time by such
Committee, and was subject topro-ration for partial service in 2016.Committee. In
2018,2020, Dr. Macrae’s base salary was
$636,480$681,797 and his target cash bonus was 60% of his base salary.
Dr. Macrae’s employment agreement also provided for a $200,000
sign-on bonus, which was subject to repayment in the event Dr. Macrae’s employment was terminated with cause or by him without good reason within one year of his appointment, as well as an initial equity grant of stock options under the 2013 Plan to acquire 700,000 shares of our common stock, which is vesting on the same terms as other discretionary awards under the 2013 Plan. Dr. Macrae is also entitled to be nominated for election to our Board of Directors for so long as his employment agreement is in effect.
Although Dr. Macrae’s employment agreement includes certain benefits payable to him in connection with separation from service, such terms were waived by Dr. Macrae in connection with the March 2017 adoption of the Severance Plan, as amended and restated in February 2019 by the Amended Severance Plan, which now governs his severance benefits. The terms of the Severance Plan and Amended Severance Plan are described below under “—Executive Severance Plan.”
Executive Vice President Employment
and Other Agreements
Ms. Yi
Mr. Lee
Effective February 28, 2017,October 31, 2019, we entered into an employment agreement with Kathy Y. Yi, which setsMr. Lee setting forth the terms and conditions of herhis employment as an Executive Vice President and Chief Financial Officer. Pursuant to herhis employment agreement, Ms. Yi will receive aMr. Lee received an initial base salary (initially $350,000of $435,000 per year)year, and an annual cash bonus based upon our company’s and herhis individual achievement of specified objectives under our Incentive Plan with a target cash bonus based on a percentage of her40% of his base salary. Mr. Lee’s base salary (initially 35%). Ms. Yi’sand target bonus percentage were subject to annual review by the Compensation Committee and adjustment from time to time by such Committee, and his salary was subject to proration for partial service in 2019. In 2020, Mr. Lee’s base salary was $438,262 and his target cash bonus was 40% of his base salary. Mr. Lee resigned effective February 1, 2021.
Mr. Lee’s employment agreement also provided for a $200,000 sign-on bonus, which was subject to repayment in the event Mr. Lee’s employment was terminated with cause or by him without good reason within one year of his appointment, as well as an initial equity grant of stock options under the 2018 Plan to acquire 262,500 shares of our common stock and a restricted stock award covering 43,750 shares of our common stock, both of which vest on the same terms as other discretionary awards under the 2018 Plan.
In addition, Mr. Lee’s employment agreement provided that he would be eligible to receive certain severance benefits under the Amended Severance Plan, which is described below. As Mr. Lee voluntarily resigned effective February 1, 2021, he did not receive any payments or benefits under the Amended Severance Plan.
In connection with Mr. Lee’s resignation, on January 22, 2021, we entered into a letter agreement with Mr. Lee pursuant to which Mr. Lee agreed to provide services to us in an independent contractor role for the period from February 2, 2021 through February 26, 2021. Mr. Lee’s compensation for this independent contractor role consisted solely of a payment of $215,000, payable in one lump sum on February 26, 2021, which was deemed to be the annual cash bonus payable to him under the Incentive Plan for 2020.
Mr. Loeb
Effective July 29, 2019, we entered into an employment agreement with Mr. Loeb setting forth the terms and conditions of his employment as an Executive Vice President and General Counsel. Pursuant to his employment agreement, Mr. Loeb received an initial base salary of $400,000 per year, and an annual cash bonus based upon our company’s and his individual achievement of specified objectives under our Incentive Plan with a target cash bonus of 40% of his base salary. Mr. Loeb’s base salary and target bonus percentage are subject to annual review by the Compensation Committee and
adjustment from time to time by such Committee, and his salary was subject to proration for partial service in 2019. In 2020, Mr. Loeb’s base salary was $406,000 and his target cash bonus was 40% of his base salary.
Mr. Loeb’s employment agreement also provided for an initial equity grant of stock options under the 2018 Plan to acquire 250,000 shares of our common stock, which vests on the same terms as other discretionary awards under the 2018 Plan.
In addition, Mr. Loeb’s employment agreement provides that he will be eligible to receive certain severance benefits under the Amended Severance Plan, which is described below.
Mr. McClung
Effective May 1, 2020, we entered into an employment agreement with Mr. McClung setting forth the terms and conditions of his employment as an Executive Vice President, Chief Business Officer. Pursuant to his employment agreement, Mr. McClung received an initial base salary of $415,000 per year, and an annual cash bonus based upon our company’s and his individual achievement of specified objectives under our Incentive Plan with a target cash bonus of 40% of his base salary. Mr. McClung’s base salary and target bonus percentage are subject to annual review by the Compensation Committee and adjustment from time to time by such Committee, and were alsohis salary was subject topro-ration proration for partial service in 2017. 2020.
Mr. McClung’s employment agreement also provided for an initial equity grant of stock options under the 2018 Plan to acquire 200,000 shares of our common stock, which vests on the same terms as other discretionary awards under the 2018 Plan and a restricted stock award covering 100,000 shares of our common stock, both of which vest on the same terms as other discretionary awards under the 2018 Plan.
In addition, Mr. McClung’s employment agreement provides that he will be eligible to receive certain severance benefits under the Amended Severance Plan, which is described below.
Dr. Ramelmeier
Effective January 1, 2018,
Ms. Yi’swe entered into an employment agreement with Dr. Ramelmeier setting forth the terms and conditions of his employment as a Senior Vice President, Technical Operations. Effective September 16, 2019, Dr. Ramelmeier was promoted to Executive Vice President, Technical Operations and his base salary
pursuant to his employment agreement was
$404,833increased to $400,000 per year and
herhis target cash bonus was
35%increased to 40% of
herhis base salary.
Ms. Yi’s Dr. Ramelmeier’s base salary and target bonus percentage are subject to annual review by the Compensation Committee and adjustment from time to time by such Committee. In 2020, Dr. Ramelmeier’s base salary was $416,000 and his target cash bonus was 40% of his base salary.
Dr. Ramelmeier’s employment agreement also provided for an initial equity grant of stock options under the 2013 Plan to acquire
200,000120,000 shares of our common stock, which
is vestingvests on the same terms as other discretionary awards under the 2013 Plan.
Although Ms. Yi’s In December 2019, the Compensation Committee approved a special cash bonus of $100,000 to be paid to Dr. Ramelmeier in four equal payments of $25,000 in July 2020, December 2020, July 2021 and December 2021.
In addition, Dr. Ramelmeier’s employment agreement
includesprovides that he will be eligible to receive certain severance benefits
payable to her in connection with separation from service, such terms were waived by Ms. Yi in connection with the March 2017 adoption of the Severance Plan, as amended and restated in February 2019 byunder the Amended Severance Plan, which
now governs her severance benefits. The terms of the Severance Plan and Amended Severance Plan areis described
below under “—Executive Severance Plan.”50
below.
In connection with our acquisition of
TxCell,Sangamo France, we entered into an employment agreement with Mr. Boissel in October 2018, setting forth the terms and conditions of his employment as our Executive Vice President, Corporate Strategy. Mr. Boissel was the former Chief Executive Officer of
TxCell.Sangamo France. Pursuant to his employment agreement, Mr. Boissel
will receivereceived an initial base salary of $480,000 per year, and an annual cash bonus based upon our company’s and his individual achievement of specified objectives under our Incentive Plan with a target cash bonus of 40% of his base salary. Under the terms of his employment agreement, Mr. Boissel was employed on a part-time basis through December 31, 2018, during which time his salary was at a rate of $140,000 per year. Mr. Boissel continued to receive compensation under his management contract with
TxCellSangamo France through December 31, 2018. Mr. Boissel’s base salary and target bonus percentage
arewere subject to annual review by the Compensation Committee and adjustment from time to time by such Committee, and his salary was subject to
pro-ration proration for partial service and part-time basis in 2018.
In 2020, Mr. Boissel’s base salary was $502,944 and his target cash bonus was 40% of his base salary.
Mr. Boissel’s employment agreement also provided for a $100,000sign-on bonus, which iswas subject to repayment in the event Mr. Boissel’s employment iswas terminated with cause or by him without good reason within one year of his appointment, as well as an initial equity grant of stock options under the 2018 Plan to acquire 215,000 shares of our common stock, which iswas vesting on the same terms as other discretionary awards under the 2018 Plan. Further, in connection with our acquisition of TxCell,Sangamo France, Mr. Boissel, as an employee of TxCell,Sangamo France, was granted a stock option under the 2018 Plan to acquire 68,523 shares of common stock with an exercise price equal to the closing price per share on the grant date. The stock option iswas subject to a four-year vesting schedule with 25% of the shares subject to the option vesting upon
Mr. Boissel’s completion of one year of service measured from the option grant date and the balance of the shares vesting monthly thereafter for the next three
years.years, subject to Mr. Boissel’s continuous service through each applicable vesting date.
In addition, Mr. Boissel’s employment agreement provided that he
will be eligible to receive certain severance benefits under the Severance Plan, as amended and restated in February 2019 by the Amended Severance Plan, which is described below.Senior Vice President Employment Agreements
Dr. Conner
Effective November 1, 2016, we entered into an employment agreement with Dr. Conner, which sets forth the terms and conditions of his employment as Chief Medical Officer. Pursuant to his employment agreement, Dr. Conner will receive a base salary (initially $400,000 per year), and an annual cash bonus based upon our company’s and his individual achievement of specified objectives under our Incentive Plan with a target cash bonus based on a percentage of his base salary (initially 35%). Dr. Conner’s base salary and target bonus percentage are subject to annual review by the Compensation Committee and adjustment from time to time by such Committee, and his salary was subject topro-ration for partial service in 2016. Dr. Conner was not eligible for a target cash bonus in 2016. In 2018, Dr. Conner’s base salary was $408,000 and his target cash bonus was 35% of his base salary.
Dr. Conner’s employment agreement also provided for a $100,000sign-on bonus, which was subject to repayment in the event Dr. Conner’s employment was terminated with cause or by him without good reason within one year of his appointment, as well as an initial equity grant of stock options under the 2013 Plan to acquire 200,000 shares of our common stock, which is vesting on the same terms as other discretionary awards under the 2013 Plan.
Although Dr. Conner’s employment agreement includes certain severance benefits payable to him in connection with separation from service, such terms were waived by Dr. Conner in connection with the March 2017 adoption of the Severance Plan, as amended and restated in February 2019 by the Amended Severance Plan, which now governs his severance benefits. The terms of the Severance Plan and Amended Severance Plan are described below under “—Executive Severance Plan.”
51
Ms. Turner
In January 2018, we entered into an employment agreement with Ms. Turner setting forth the terms and conditions of her employment as a Senior Vice President and General Counsel and Secretary. Pursuant to her employment agreement, Ms. Turner received an initial base salary of $400,000 per year, and an annual cash bonus based upon our company’s and her individual achievement of specified objectives under our Incentive Plan with a target cash bonus of 35% of her base salary. Ms. Turner’s base salary and target bonus percentage were subject to annual review by the Compensation Committee and adjustment from time to time by such Committee, and her salary was subject topro-ration for partial service in 2018.
Ms. Turner’s employment agreement also provided for a $150,000sign-on bonus, which was subject to repayment in the event Ms. Turner’s employment was terminated with cause or by her without good reason within one year of her appointment, as well as an initial equity grant of stock options under the 2013 Plan to acquire 200,000 shares of our common stock, which was vesting on the same terms as other discretionary awards under the 2013 Plan (and which has since lapsed in accordance with its terms following her voluntary resignation).
Ms. Turner’s employment agreement provided that she was eligible to receive certain severance benefits under the Severance Plan, as amended and restated in February 2019 by the Amended Severance Plan. As Ms. Turner voluntarily resigned in March 2019, she did not receive any benefits under the Amended Severance Plan.
Executive Severance Plan
Executive Severance Plan in Effect as of December 31, 2018
In March 2017, the Compensation Committee adopted the Severance Plan, to provide severance benefits to certain of our executive officers and other key employees whose employment terminates under certain prescribed circumstances. Eligible employees under the Severance Plan include our named executive officers Drs. Macrae and Conner, Ms. Yi and Mr. Boissel. We have excluded Ms. Turner from the below as she voluntarily resigned effective March 2019 and is not eligible to receive any severance benefits.
Under the Severance Plan, which replaced theis described below. Mr. Boissel resigned effective July 31, 2020 and pursuant to a Separation Agreement will receive severance arrangement set forth in Dr. Macrae’s employment agreement, Dr. Macrae would have beenbenefits of $628,680 over a 15-month period and is eligible to receive the following severance benefits: (a) cash equal to the sumfor reimbursement of (i) 18 months of his base salary and (ii) his target bonus for the year of termination, payable over 12 months, reimbursement for his health care coverage costs under COBRA for 12 months, and full acceleration of his outstanding equity awards, and any outstanding options as so accelerated would have remained exercisable15 months. Mr. Boissel also received a $25,000 payment for a period of 12 months following termination, in the event of an involuntary termination duringthe 12-month period following a change in control ofconsulting services rendered to the Company or the Change in Control Period, or (b) cash equalsubsequent to 12 months of his base salary, payable over 12 months, and COBRA reimbursement for 12 months, if he had an involuntary termination other than during the Change in Control Period. These cash severance benefits under the Severance Plan were identical to the severance benefits he was eligible to receive under his employment agreement; however, under his employment agreement, only 50% of the shares subject to any outstanding equity award accelerated if the change in control was within two years following the effective date of the employment agreement and 100% of the shares accelerated if the change in control was more than two years after the effective date.
Under the Severance Plan, for which Mr. Boissel was eligible to receive benefits and which replaced the severance arrangements set forth in Dr. Conner’s, Ms. Yi’s and Ms. Turner’s respective employment agreements, Mr. Boissel, Dr. Conner, Ms. Yi and Ms. Turner would have been eligible to receive the following severance benefits: (a) cash equal to the sum of (i) 12 months of base salary and (ii) target bonus for the year of termination, payable over 12 months, reimbursement for health care coverage costs under COBRA for 12 months, and accelerated vesting of all outstanding equity awards, and any outstanding options as so accelerated
52
resignation.
would have remained exercisable for a period of 12 months following termination, in the event of an involuntary termination during the Change in Control Period; or (b) cash equal to nine months of base salary, payable over nine months, and COBRA reimbursement for nine months, if there had been an involuntary termination other than during the Change in Control Period. These cash severance benefits under the Severance Plan were similar to the severance benefits Dr. Conner and Ms. Yi were eligible to receive under their respective employment agreements; however, under the employment agreements, only 50% of the shares subject to any outstanding equity award accelerated if the change in control was within two years following the effective date of the employment agreement and 100% of the shares accelerated if the change in control was more than two years after the effective date, and (b) the base salary and COBRA reimbursement continuation period was six months under the employment agreements (instead of 12 months in the event of an involuntary termination during the Change in Control Period or nine months in the event of an involuntary termination other than during the Change in Control Period, under the Severance Plan, as described above).
Current
In February 2019, the Compensation Committee of the Board adopted the Amended Severance Plan, which amended and restated the Severance Plan.
Under the Amended Severance Plan, Dr. Macrae is eligible to receive the following severance benefits: (a) cash equal to the sum of (i) 18 months of his base salary and (ii) 1/12 of the target bonus for the year of termination multiplied by 18, payable over 12 months, reimbursement for his health care coverage costs under COBRA for 18 months, and full acceleration of his outstanding equity awards, and any outstanding options as so accelerated will remain exercisable for a period of 12 months following termination, in the event of an involuntary termination during the Change in Control Period or (b) cash equal to 18 months of his base salary, payable over 18 months, and COBRA reimbursement for 18 months, if he has an involuntary termination other than during the Change in Control Period.
Under the Amended Severance Plan,
Ms. YiMr. Loeb, Mr. McClung and Dr. Ramelmeier are eligible (and had they not resigned, Mr. Lee and Mr. Boissel
are eligiblewould have been eligible) to receive the following severance benefits: (a) cash equal to the sum of (i) 15 months of base salary and (ii) 1/12 of the target bonus for the year of termination multiplied by 15, payable over 15 months, reimbursement for health care coverage costs under COBRA for 15 months, and accelerated vesting of all outstanding equity awards, and any outstanding options as so accelerated will remain exercisable for a period of 12 months following termination, in the event of an involuntary termination during the Change in Control Period; or (b) cash equal to 15 months of base salary, payable over 15 months, and COBRA reimbursement for 15 months, if there is an involuntary termination other than during the Change in Control Period.
Under the Amended Severance Plan, Dr. Conner and Ms. Turner are eligible to receive the following severance benefits: (a) cash equal to the sum of (i) 12 months of base salary and (ii) 1/12 of the target bonus for the year of termination multiplied by 12, payable over 12 months, reimbursement for health care coverage costs under COBRA for 12 months, and accelerated vesting of all outstanding equity awards, and any outstanding options as so accelerated will remain exercisable for a period of 12 months following termination, in the event of an involuntary termination during the Change in Control Period; or (b) cash equal to 12 months of base salary, payable over 12 months, and COBRA reimbursement for 12 months, if there is an involuntary termination other than during the Change in Control Period.
If any of the severance benefits under the Amended Severance Plan would constitute a “parachute payment” within the meaning of Section 280G of the Code, such payments are subject to reduction to the extent doing so would put the recipient in a better
after-tax position after taking into account any excise tax that may be incurred under Code Section 4999 in connection with any change in control of the Company or subsequent termination of employment.
53
The Compensation Committee of the Board of Directors, as the administrator of the 2013 Plan, has the authority to provide that any outstanding options held by the Chief Executive Officer or any other executive officer or any other unvested equity award made to such individual under the 2013 Plan will vest on an accelerated basis in connection with certain changes in control of the Company or the subsequent termination of the officer’s employment following the change in control event. In addition, all outstanding options and RSUs under the 2013 Plan will immediately vest upon a change in control, to the extent not assumed or continued in effect by the successor entity or replaced with an incentive compensation program that preserves the intrinsic value of the award at that time and provides for the subsequent vesting and concurrent payout of that value in accordance with the
pre-existing vesting schedules for those awards.
The Board of Directors, or any committee properly constituted thereof, as the administrator of the 2018 Plan, has the authority to accelerate the time at which an equity award made under the 2018 Plan may first be exercised or vest on an accelerated basis in connection with certain changes in control of the Company. In addition, all outstanding options and RSUs under the 2018 Plan will immediately vest upon a change in control, to the extent not assumed or continued in effect by the successor entity or replaced with a similar incentive compensation program, for which the Board of Directors shall set the terms of any assumption, continuation or substitution.
Potential Payments Upon Termination or Change in Control as of December 31,
20182020
The charts below quantify the potential payments our named executive officers,
other than Mr. Boissel, would receive under various
scenarios under the Severance Plan.scenarios. The value attributed to the accelerated vesting of equity awards represents the intrinsic value of each stock option or RSU award vesting on an accelerated basis in connection with the identified triggering event, which is assumed to occur on December 31,
2018.2020. The intrinsic value is calculated by multiplying (i) the aggregate number of shares that vest on an accelerated basis by (ii) the amount by which the
$11.48$15.61 closing selling price per share of our common stock on December 31,
2018,2020, exceeds the exercise price
payable or other issue price (if any)
payable per vested share.
Because Mr. Boissel resigned effective July 31, 2020, he was not eligible for any potential payments or benefits under any of the various scenarios
below as of December 31, 2020, and he did not receive any payments or benefits under our Amended Severance Plan. Mr. Boissel received payments pursuant to a Separation Agreement which are described and quantified above under “—Executive Vice President Employment and Other Agreements—Mr. Boissel.”
Quantification of Benefits Upon Involuntary Termination in the Absence of a Change in Control
The chart below quantifies the compensation each named executive officer would have received had the officer’s employment terminated without cause in the absence of a change in control of the Company but under circumstances entitling the officer to severance benefits under the
Amended Severance Plan.
| | | | | | | | |
Name | | Cash
Severance ($) (1) | | | COBRA ($) | |
Alexander D. Macrae | | | 636,480 | | | | 17,392 | |
Kathy Y. Yi | | | 303,625 | | | | 13,044 | |
Heather D. Turner | | | 300,000 | | | | 21,755 | |
Stéphane Boissel | | | 360,000 | | | | 7,430 | |
Edward R. Conner, M.D. | | | 306,000 | | | | 2,488 | |
(1) | Cash severance upon termination in the absence of a change in control is payable in a series of successive equal monthly installments over a period ranging from nine to 12 months.
|
54
| | | | | | | | | | | | | | |
| Name | | Cash Severance ($) (1) | | COBRA ($) |
| Alexander D. Macrae | | 1,022,695 | | | 28,144 | |
| Sung H. Lee (2) | | 547,827 | | | 39,114 | |
| Gary H. Loeb | | 507,500 | | | 11,604 | |
| D. Mark McClung | | 518,750 | | | 2,578 | |
| R. Andrew Ramelmeier | | 520,000 | | | 28,176 | |
___________________
(1)Cash severance upon termination in the absence of a change in control is payable in a series of successive equal monthly installments over a period ranging from 15 months (named executive officers other than CEO) to 18 months (CEO).
(2)Mr. Lee resigned effective February 1, 2021 and did not receive any severance benefits. On January 22, 2021, we entered into a letter agreement with Mr. Lee, pursuant to which Mr. Lee agreed to provide services to us in an independent contractor role for the period from February 2, 2021 through February 26, 2021. Mr. Lee’s compensation for this independent contractor role consisted solely of a payment of $215,000, payable in one lump sum on February 26, 2021, which was deemed to be the annual cash bonus payable to him under the Incentive Plan for 2020.
Benefits Upon Involuntary Termination in Connection With a Change in Control
The chart below quantifies the payments our named executive officers would each have received had their employment terminated without cause or with good reason in connection with a change in control under circumstances entitling them to severance benefits under the
Amended Severance Plan.
| | | | | | | | | | | | | | | | | | | | |
Name | | Cash
Severance ($) (1) | | | Target
Bonus ($) (2) | | | Accelerated
Vesting of
Equity
Awards ($) (3) | | | COBRA ($) | | | | |
Alexander D. Macrae | | | 954,720 | | | | 381,888 | | | | 3,015,495 | | | | 17,392 | | | | | |
Kathy Y. Yi | | | 404,833 | | | | 141,692 | | | | 833,985 | | | | 17,392 | | | | | |
Heather D. Turner | | | 400,000 | | | | 140,000 | | | | - | | | | 29,006 | | | | | |
Stéphane Boissel (4) | | | 480,000 | | | | - | | | | 113,409 | | | | 9,906 | | | | | |
Edward R. Conner, M.D. | | | 408,000 | | | | 142,800 | | | | 863,821 | | | | 3,317 | | | | | |
(1) | Cash severance upon termination in connection with a change in control is payable in a series of successive equal monthly installments over 12 months.
|
(2) | Target bonus represents the amount of severance benefit that an executive is entitled to payable in a series of successive equal monthly installments over 12 months.
|
(3) | No value is included in this table with respect to the accelerated vesting of options for which the exercise price was in excess of the closing price of our common stock on December 31, 2018.
|
(4) | Mr. Boissel joined the Company in October 2018 on a part-time basis through December 31, 2018 and was not eligible to participate in the annual performance bonus program during 2018.
|
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | | Cash
Severance ($) (1) | | Target
Bonus ($) (2) | | Accelerated
Vesting of
Equity
Awards ($) (3) | | COBRA ($) |
| Alexander D. Macrae | | 1,022,695 | | | 613,617 | | | 7,873,140 | | | 28,144 | |
| Sung H. Lee | | 547,827 | | | 219,131 | | | 3,126,098 | | | 39,114 | |
| Gary H. Loeb | | 507,500 | | | 203,000 | | | 2,426,502 | | | 11,604 | |
| D. Mark McClung | | 518,750 | | | 207,500 | | | 4,412,000 | | | 2,578 | |
| R. Andrew Ramelmeier | | 520,000 | | | 208,000 | | | 2,225,041 | | | 28,176 | |
___________________
(1)Cash severance upon termination in connection with a change in control is payable in a series of successive equal monthly installments over a period ranging from 15 months (named executive officers other than CEO) to 18 months (CEO).
(2)Target bonus represents the amount of severance benefit that an executive is entitled to payable in a series of successive equal monthly installments over a period ranging from 15 months (named executive officers other than CEO) to 18 months (CEO).
(3)No value is included in this table with respect to the accelerated vesting of options for which the exercise price was in excess of the closing price of our common stock on December 31, 2020.
Benefits Upon a Change in Control (No Termination)
The chart below quantifies the value of the accelerated equity the named executive officers are entitled to receive under the 2013 Plan and 2018 Plan upon a change in control of the Company in which their outstanding equity awards are not assumed or otherwise continued in effect, terminated or
cancelledcanceled in connection therewith.
| | | | | | | | |
Name | | Accelerated
Vesting of
Equity
Awards ($) | |
Alexander D. Macrae | | 7,873,140 | 3,015,495 | |
Kathy Y. Yi
Sung H. Lee | | 3,126,098 | 833,985 | |
Heather D. Turner
Gary H. Loeb | | 2,426,502 | - | |
Stéphane Boissel
D. Mark McClung | | 4,412,000 | 113,409 | |
Edward R. Conner, M.D.
Andrew Ramelmeier | | 2,225,041 | 863,821 | |
Benefits upon Death or Disability
Under the 2013 Plan, if a named executive officer’s service with us is terminated as a result of death or permanent disability, or a named executive officer dies or becomes permanently disabled during the three months after a termination other than for misconduct, the period of time in which an option may be exercised following termination shall be the earlier of 12 months following the date of such named executive officer’s death or the expiration date of the option, whichever is earlier, rather than the standard three-month post-termination exercise period provided for in the 2013 Plan or the 2018 Plan.
Potential Payments Upon Termination or Change in Control Under Amended Severance Plan
As discussed above, in February 2019, the Compensation Committee of the Board adopted the Amended Severance Plan, which amended and restated the Severance Plan. To provide meaningful disclosure to investors,
55
we estimate in the charts below the amount of potential compensation to our named executed officers under various scenarios under the Amended Severance Plan, assuming the applicable event occurred on December 31, 2018, the last business day of the Company’s last fiscal year, and the Amended Severance Plan had instead been in effect on December 31, 2018. The value attributed to the accelerated vesting of equity awards represents the intrinsic value of each stock option or RSU award vesting on an accelerated basis in connection with the identified triggering event, which is assumed to occur on December 31, 2018. The intrinsic value is calculated by multiplying (i) the aggregate number of shares that vest on an accelerated basis by (ii) the amount by which the $11.48 closing selling price per share of our common stock on December 31, 2018, exceeds the exercise price payable or other issue price (if any) per vested share.
Quantification of Benefits Upon Involuntary Termination in the Absence of a Change in Control
The chart below quantifies the compensation each named executive officer would have received had the officer’s employment terminated without cause in the absence of a change in control of the Company but under circumstances entitling the officer to severance benefits under the Amended Severance Plan.
| | | | | | | | | | | | |
Name | | Cash
Severance ($) (1) | | | COBRA ($) | | | | |
Alexander D. Macrae | | | 954,720 | | | | 26,088 | | | | | |
Kathy Y. Yi | | | 506,841 | | | | 21,740 | | | | | |
Heather D. Turner | | | 400,000 | | | | 29,006 | | | | | |
Stéphane Boissel | | | 600,000 | | | | 12,383 | | | | | |
Edward R. Conner, M.D. | | | 408,000 | | | | 3,317 | | | | | |
(1) | Cash severance upon termination in the absence of a change in control is payable in a series of successive equal monthly installments over a period ranging from 12 to 18 months.
|
Benefits Upon Involuntary Termination in Connection With a Change in Control
The chart below quantifies the payments our named executive officers would each have received had their employment terminated without cause or with good reason in connection with a change in control under circumstances entitling them to severance benefits under the Amended Severance Plan.
| | | | | | | | | | | | | | | | | | | | |
Name | | Cash
Severance ($) (1) | | | Target
Bonus ($) (2) | | | Accelerated
Vesting of
Equity
Awards ($) (3) | | | COBRA ($) | | | | |
Alexander D. Macrae | | | 954,720 | | | | 572,832 | | | | 3,015,495 | | | | 26,088 | | | | | |
Kathy Y. Yi | | | 506,841 | | | | 177,114 | | | | 833,985 | | | | 21,740 | | | | | |
Heather D. Turner | | | 400,000 | | | | 140,000 | | | | - | | | | 29,006 | | | | | |
Stéphane Boissel (4) | | | 600,000 | | | | - | | | | 113,409 | | | | 12,383 | | | | | |
Edward R. Conner, M.D. | | | 408,000 | | | | 142,800 | | | | 863,821 | | | | 3,317 | | | | | |
(1) | Cash severance upon termination in connection with a change in control is payable in a series of successive equal monthly installments over a period ranging from 12 to 15 months.
|
(2) | Target bonus represents the amount of severance benefit that an executive is entitled to payable in a series of successive equal monthly installments over a period ranging from 12 to 15 months.
|
(3) | No value is included in this table with respect to the accelerated vesting of options for which the exercise price was in excess of the closing price of our common stock on December 31, 2018.
|
(4) | Mr. Boissel joined the Company in October 2018 on a part-time basis through December 31, 2018 and was not eligible to participate in the annual performance bonus program during 2018.
|
56
Benefits Upon a Change in Control (No Termination)
The chart below quantifies the value of the accelerated equity the named executive officers are entitled to receive under the 2013 Plan and 2018 Plan upon a change in control of the Company in which their outstanding equity awards are not assumed or otherwise continued in effect, terminated or cancelled in connection therewith.
| | | | | | | | |
Name
| | Accelerated Equity ($) | | | | |
Alexander D. Macrae
| | | 3,015,495 | | | | | |
Kathy Y. Yi
| | | 833,985 | | | | | |
Heather D. Turner
| | | - | | | | | |
Stéphane Boissel
| | | 113,409 | | | | | |
Edward R. Conner, M.D.
| | | 863,821 | | | | | |
Benefits upon Death or Disability
Under the 2013 Plan, if a named executive officer’s service with us is terminated as a result of death or permanent disability, or a named executive officer dies or becomes permanently disabled during the three months after a termination other than for misconduct, the period of time in which an option may be exercised following termination shall be the earlier of 12 months following the date of such named executive officer’s death or the expiration date of the option, whichever is earlier, rather than the standard three-month post-termination exercise period provided for in the 2013 Plan or the 2018 Plan.
CEO Pay Ratio Disclosure
Under SEC rules, we are required to calculate and disclose the annual total compensation of our median employee, as well as the ratio of the annual total compensation of our median employee as compared to the annual total compensation of our President and Chief Executive Officer, Dr. Macrae,
(our “CEO”),our CEO, for our last fiscal year,
(“or the CEO Pay
Ratio”).Ratio. To identify our median employee, we used the following methodology:
•To determine our total population of employees, we included all of our employees as of November 15, 20182020 regardless of their full- or part-time schedule or anticipated employment duration.
•To identify our median employee from our employee population, we calculated the aggregate amount of the 20182020 base compensation of each of our employees excluding the CEO using a reasonable estimate of the hours worked during 20182020 for our hourly employees and actual salary paid for our remaining employees, target 20182020 bonus, and the grant date fair value of equity awards granted in fiscal 20182020 (using the same methodology we use for estimating the value of the equity awards granted to our named executive officers and reported in our Summary Compensation Table).
•In making this determination, we annualized the base compensation, target bonus and other cash incentive compensation of those permanent employees who were employed by us for less than the entire fiscal year.
Using this approach, we determined our median
employee. Our determination of the median employee
yielded two median employees because, after excluding the CEO, we had an even number of employees. After identifying the two median employees, weand determined their annual total compensation in accordance with the requirements of the Summary Compensation
Table and, in order to utilize a more conservative approach, selected the employee with the lower annual total compensation.Table.
Accordingly, for fiscal
2018,2020, the median of the annual total compensation of our employees (other than our CEO) was
$173,399$174,051 and the annual total compensation of our CEO, as reported in the Summary Compensation Table included in this Proxy Statement, was
$4,104,428.$3,835,030. Based on this information, the ratio of the annual total compensation of our CEO to the median of the annual total compensation of all our employees was approximately
2422 to 1.
57
The CEO Pay Ratio above represents our reasonable estimate calculated in a manner consistent with SEC rules and applicable guidance. SEC rules and guidance provide significant flexibility in how companies identify the median employee, and each company may use a different methodology and make different assumptions particular to that company. As a result, and as explained by the SEC when it adopted these rules, in considering the pay ratio disclosure, stockholders should keep in mind that the rule was not designed to facilitate comparisons of pay ratios among different companies, even companies within the same industry, but rather to allow stockholders to better understand and assess each particular company’s compensation practices and pay ratio disclosures.
Neither the Compensation Committee nor our management used our CEO Pay Ratio measure in making compensation decisions.
Compensation Committee Report1
COMPENSATION COMMITTEE REPORT(1)
The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis contained in this Proxy Statement with management, and based on such review and such discussions, the Compensation Committee recommended to the Board of Directors that the Compensation Discussion and Analysis, as contained herein, be included in this Proxy Statement.
| | |
|
| Submitted by the Compensation |
| Committee of the |
| Board of Directors of Sangamo Therapeutics, Inc. |
|
Dr. Steven J. Mento
Dr. Roger Jeffs
Mr. Joseph S. Zakrzewski |
Ms. H. Stewart ParkerDr. Karen L. Smith |
| Mr. James R. Meyers |
1 | ___________________ (1)The material in this Compensation Committee Report is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference into any filing of Sangamo Therapeutics, Inc. under the Securities Act of 1933, as amended, or the Securities Act, or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing. |
Equity Compensation Plan Information
Committee Report is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference into any filing of Sangamo Therapeutics, Inc. under the Securities Act of 1933, as amended, or the Securities Act, or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
EQUITY COMPENSATION PLAN INFORMATION
The following table provides information as of December 31,
2018,2020, with respect to the shares of our common stock that may be issued under our existing equity compensation plans. There are no outstanding options that we have assumed in connection with our acquisition of other companies, and there are currently no assumed plans under which we can grant options.
| | | | | | | | | | | | | | | | | | | | | | | | |
| | | Column (A) | | | | | | Column (B) | | | | | | Column (C) | | | | |
Plan Category | | Number of
Securities to be
Issued Upon
Exercise of
Outstanding Options,
Restricted Stock Units
and Other Rights | | | | | | Weighted Average
Exercise Price of
Outstanding
Options | | | | | | Number of Securities
Remaining Available for
Future Issuance Under
Equity Compensation
Plans
(Excluding Securities
Reflected in Column A) | | | | |
| Equity Compensation Plans Approved by Stockholders (1) | | | 8,684,793 | | | | (2 | )(3) | | $ | 10.85 | | | | (4 | ) | | | 12,498,382 | | | | (5 | )(6) |
| Equity Compensation Plans Not Approved by Stockholders (1) | | | 364,000 | | | | (7 | ) | | | 19.92 | | | | | | | | - | | | | (6 | )(8) |
| | | | | | | | | | | | | | | | | | | | | | | | |
Total | | | 9,048,793 | | | | | | | | 11.23 | | | | | | | | 12,498,382 | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | |
(1) | The equity compensation plans approved by stockholders consists of the 2013 Plan, the 2018 Plan and the Purchase Plan. In November 2017, the Compensation Committee approved the amendment and restatement
|
58
| of the 2013 Plan to reserve an additional 1,000,000 shares of our common stock to be used exclusively for grants of awards to individuals who were not previously employees ornon-employee directors of the Company (or following a bona fide period ofnon-employment with the Company), as an inducement material to each such individual’s entry into employment with us within the meaning of Rule 5635(c)(4) of the Nasdaq Listing Rules, or Rule 5635(c)(4) (such awards, the Inducement Awards). The 2013 Plan was amended and restated by the Compensation Committee without stockholder approval pursuant to Rule 5635(c)(4). Accordingly, for purposes of this table, information with respect to Inducement Awards and the shares reserved for issuance under the 2013 Plan therefor, are included in the row in the table above with respect to equity compensation plans not approved by stockholders.
|
(2) | Includes 322,701 shares subject to RSUs that will entitle the holder to one share of common stock for each unit that vests over the holder’s period of continued service.
|
(3) | Excludes purchase rights accruing under the Purchase Plan and shares subject to outstanding options granted under the 2013 Plan as Inducement Awards. Under the Purchase Plan, each eligible employee may purchase up to 2,000 shares of common stock at semi-annual intervals on the last U.S. business day of April and October each year at a purchase price per share equal to 85% of the lower of (i) the closing selling price per share of common stock on the employee’s entry date into thetwo-year offering period in which that semi-annual purchase date occurs or (ii) the closing selling price per share on the semi-annual purchase date.
|
(4) | The calculation does not take into account the 322,701 shares of common stock subject to outstanding RSUs. Such shares will be issued at the time the RSUs vest, without any cash consideration payable for those shares.
|
(5) | Consists of shares available for future issuance under the Purchase Plan and the 2018 Plan. As of December 31, 2018, 3,006,964 shares of common stock were available for issuance under the Purchase Plan, and 9,491,418 shares of common stock were available for issuance under the 2018 Plan. The shares available for issuance under the 2018 Plan may be issued upon the exercise of stock options or stock appreciation rights granted under the plan, or those shares may be issued as stock bonuses or pursuant to RSUs that vest upon the attainment of prescribed performance milestones or the completion of designated service periods.
|
(6) | As of December 31, 2018, the maximum number of shares of common stock reserved for issuance under the 2018 Plan and the Purchase Plan was 10,418,588 shares and 4,600,000 shares, respectively. The number of shares of common stock reserved for issuance under the 2018 Plan is reduced: (i) on a1-for-1 basis for each share of common stock subject to a stock option grant or stock appreciation right award, or pursuant to a full-value award made under the 2013 Plan, and (ii) by a fixed ratio of 1.33 shares of common stock for each share of common stock issued pursuant to a full-value award made under the 2018 Plan.
|
(7) | Consists of stock options granted as Inducement Awards under the 2013 Plan.
|
(8) | As of December 31, 2018, options to purchase 364,000 shares were outstanding as Inducement Awards. All options granted as Inducement Awards have a maximum term of 10 years. As a result of the adoption of the 2018 Plan at our 2018 annual meeting of stockholders, no additional stock awards may be granted as Inducement Awards. Accordingly, for purposes of the table above, no shares remained available for issuance as Inducement Awards as of December 31, 2018.
|
59
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Column (A) | | | | | Column (B) | | | | Column (C) | | | |
| Plan Category | | Number of
Securities to be
Issued Upon
Exercise of
Outstanding Options,
RSUs
and Other Rights | | | | | Weighted-Average
Exercise Price of
Outstanding
Options | | | | Number of Securities
Remaining Available for
Future Issuance Under
Equity Compensation
Plans
(Excluding Securities
Reflected in Column A) | | | |
Equity Compensation Plans Approved by Stockholders (1) | | 14,026,226 | | | (2) | | (3) | | $ | 9.96 | | | (4) | | | 13,425,794 | | | (5) | | (6) |
Equity Compensation Plans Not Approved by Stockholders (1) | | 124,000 | | | (7) | | (8) | | 15.00 | | | | | 5,000,000 | | | (9) | | (10) |
| Total | | 14,150,226 | | | | | | 10.02 | | | | | 18,425,794 | | | | |
Report___________________
(1)The equity compensation plans approved by stockholders consist of the Audit2013 Plan, the 2018 Plan and the 2010 ESPP. In November 2017, the Compensation Committee approved the amendment and restatement of the Board2013 Plan to reserve an additional 1,000,000 shares of Directors1our common stock to be used exclusively for grants of awards to individuals who were not previously employees or non-employee directors of the Company (or following a bona fide period of non-employment with the Company), as an inducement material to each such individual’s entry into employment with us within the meaning of Rule 5635(c)(4) of the Nasdaq Listing Rules, or Rule 5635(c)(4) (such awards, the Inducement Awards). The 2013 Plan was amended and restated by the Compensation Committee without stockholder approval pursuant to Rule 5635(c)(4). Accordingly, for purposes of this table, information with respect to Inducement Awards and the shares reserved for issuance under the 2013 Plan therefor, are included in the row in the table above with respect to equity compensation plans not approved by stockholders.
(2)Includes 2,671,528 shares subject to RSUs that will entitle the holder to one share of common stock for each unit that vests over the holder’s period of continuous service.
(3)Excludes purchase rights accruing under the 2010 ESPP and shares subject to outstanding options granted under the 2013 Plan as Inducement Awards (which Inducement Awards are included in the row in the table above with respect to equity compensation plans not approved by stockholders). Under the 2010 ESPP, each eligible employee may purchase up to 2,000 shares of common stock at semi-annual intervals on the last U.S. business day of April and October each year at a purchase price per share equal to 85% of the lower of (i) the closing selling price per share of common stock on the employee’s entry date into the two-year offering period in which that semi-annual purchase date occurs or (ii) the closing selling price per share on the semi-annual purchase date.
(4)The calculation does not take into account the 2,671,528 shares of common stock subject to outstanding RSUs. Such shares will be issued at the time the RSUs vest, without any cash consideration payable for those shares.
(5)Consists of shares available for future issuance under the 2010 ESPP and the 2018 Plan. As of December 31, 2020, 2,483,218 shares of common stock were available for issuance under the 2010 ESPP, and 10,942,576 shares of common stock were available for issuance under the 2018 Plan.
(6)As of December 31, 2020, the maximum aggregate number of shares of common stock reserved for issuance under the 2018 Plan and the 2010 ESPP was 21,124,346 shares and 4,600,000 shares, respectively. This maximum aggregate share reserve is not the same as the shares available for future issuance, which is described in Footnote 5. As of December 31, 2020, the aggregate number of shares of our common stock that may be issued under the 2018 Plan will not exceed the sum of (i) 1,703,964 shares (the number of shares that were available for grant under the 2013 Plan as of immediately prior to the effective date of the 2018 Plan); (ii) 8,800,000 shares (the number of additional shares that were reserved as of the effective date of the 2018 Plan); (iii) 9,900,000 shares (the number of additional shares that were reserved as of the approval of the amendment to the 2018 Plan); and (iv) any returning shares subject to outstanding awards granted under the Prior Plans as such shares become available from time to time due to expiration or termination of such awards, settlement of such awards in cash or forfeitures or repurchases. The number of shares of our common stock available for issuance under the 2018 Plan is increased by: (i) one share for each Prior Plans’ returning share of common stock or 2018 Plan returning share of common stock subject to a stock option grant or stock appreciation right award and (ii) 1.33 shares for each Prior Plans’ returning share of common stock or 2018 Plan returning share of common stock subject to a full value award. The number of shares of common stock reserved for issuance under the 2018 Plan is reduced: (i) on a 1-for-1 basis for each share of common stock subject to a stock option grant or stock appreciation right award, or pursuant to a full-value award made under the 2018 Plan, and (ii) by a fixed ratio of 1.33 shares of common stock for each share of common stock issued pursuant to a full-value award made under the 2018 Plan.
(7)Consists of stock options granted as Inducement Awards under the 2013 Plan. As of December 31, 2019, no shares remained available for issuance as Inducement Awards under the 2013 Plan.
(8)Excludes purchase rights accruing under the 2020 ESPP. Under the 2020 ESPP, each eligible employee may purchase up to 5,000 shares of common stock at semi-annual intervals on the last trading day of May and November each year at a purchase price per share equal to 85% of the lower of (i) the closing selling price per share of common stock on the employee’s entry date into the two-year offering period in which that semi-annual purchase date occurs or (ii) the closing selling price per share on the semi-
annual purchase date. As described in Proposal No. 3, the 2020 ESPP was approved by our Compensation Committee, subject to approval by our stockholders, and accordingly, no purchase rights may be exercised under the 2020 ESPP unless Proposal No. 3 is approved by our stockholders.
(9)As of December 31, 2020, the maximum aggregate number of shares of common stock reserved for issuance under the 2020 ESPP was 5,000,000 shares. As described in Proposal No. 3, the 2020 ESPP was approved by our Compensation Committee, subject to approval by our stockholders, and accordingly, no purchase rights may be exercised under the 2020 ESPP unless Proposal No. 3 is approved by our stockholders.
(10)As of December 31, 2020, options to purchase 124,000 shares were outstanding as Inducement Awards. All options granted as Inducement Awards have a maximum term of 10 years. As a result of the adoption of the 2018 Plan at our 2018 annual meeting of stockholders, no additional stock awards may be granted as Inducement Awards. Accordingly, for purposes of the table above, no shares remained available for issuance as Inducement Awards as of December 31, 2020.
REPORT OF THE AUDIT COMMITTEE OF THE BOARD OF DIRECTORS(1)
The information contained under the heading “Report of the Audit Committee of the Board of Directors” in this Proxy Statement shall not be deemed to be “soliciting material” or to be “filed” with the
Securities and Exchange Commission,SEC, nor shall such information be incorporated by reference into any filing of the Company with the
Securities and Exchange Commission,SEC, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing, or subject to the liabilities of Section 18 of the Exchange Act.
The Audit Committee has reviewed and discussed the audited financial statements for the fiscal year ended December 31,
20182020 with the management of the Company.
The Audit Committee has discussed with Ernst & Young LLP, the independent registered public accounting firm that audited our financial statements for the fiscal year ended December 31,
2018,2020, the matters required to be discussed by
the applicable requirements of the Public Company Accounting Oversight Board,
(PCAOB) Auditing Standard No. 1301, Communications with Audit Committees,” or
any successor standard.PCAOB, and the SEC.
The Audit Committee has received the written disclosures and the letter from Ernst & Young LLP required by applicable requirements of the PCAOB regarding Ernst & Young LLP’s communications with the Audit Committee concerning independence, and has discussed with Ernst & Young LLP its independence from the Company.
Based on the review of the audited financial statements and the discussions noted above, the Audit Committee recommended to our Board of Directors that the audited financial statements be included in our Annual Report on
Form 10-K for the fiscal year ended December 31,
20182020 for filing with the
Securities and Exchange Commission.SEC. | | |
|
Submitted by the Audit Committee of the
Board of Directors of Sangamo Therapeutics, Inc.
|
|
| Mr. Robert F. Carey |
| Mr. James R. Meyers |
| Ms. Saira Ramasastry |
Mr. Joseph S. Zakrzewski |
| 1 | The material in this Report of the Audit Committee is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference into any filing of Sangamo Therapeutics, Inc. under the Securities Act or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
|
60
___________________
(1)The material in this Report of the Audit Committee is not “soliciting material,” is not deemed “filed” with the SEC and is not to be incorporated by reference into any filing of Sangamo Therapeutics, Inc. under the Securities Act or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Policy and Procedures for Review of Related Party Transactions
Consistent with the requirement under Nasdaq listing rules, the Audit Committee of the Board of Directors is responsible for reviewing and providing oversight of all related party transactions as defined under SEC rules and regulations. While we do not have a formal written policy or procedure for the review, approval or ratification of related party transactions, the charter of the Audit Committee requires that the Audit Committee approve all related party transactions as defined under SEC rules and regulations. In making its decision to approve or ratify a related party transaction, the Audit Committee will consider the relevant facts and circumstances available and deemed relevant to the Audit Committee, including but not limited to the risks, costs and benefits to us, the terms and aggregate value of the transaction, the availability of other sources for comparable services or products, and, if applicable, the impact on a director’s independence.
Transactions with Related Persons; Indemnification
Transactions with Related Persons.Since January 1, 2018,2020, there has not been nor is there currently proposed any transaction or series of similar transactions to which we were or are to be a party in which the amount involved exceeds $120,000 and in which any director, executive officer, holder of more than 5% of our common stock, or any member of the immediate family of any of the foregoing persons, had or will have a direct or indirect material interest other than described below and other than compensation agreements and other arrangements, which are described elsewhere in this Proxy Statement.As previously disclosed, in July
In October 2018,
we acquired Sangamo France and as part of that acquisition, we entered into
a share purchase agreementliquidity agreements with certain
shareholdersholders of
TxCell,“free shares” of Sangamo France, including Mr. Boissel, one of our
named executive officers,
to purchase approximately 53% of the outstanding share capital and voting rights of TxCell in a block transaction (or an aggregate of 13,519,036 ordinary shares of TxCell). Accordingly, in October 2018, we purchased 33,000 TxCell shares from Mr. Boissel for €85,140 (or $98,762) in the closing of the block transaction. In July 2018, we also entered into arrangements with the holders of approximately 477,000 “free shares” of TxCell, including Mr. Boissel, with respect to 238,100 of such
TxCell free shares. Under
these arrangements,the liquidity agreement with Mr. Boissel, we
havehad the right to purchase such
TxCell free shares from
the holders thereofMr. Boissel and
such holders haveMr. Boissel had the right to sell to us such
TxCell free shares from time to time through
mid-2021. TheFebruary 2021 for a purchase price
for each such TxCellper free share
as set forth in the liquidity agreement. Since January 1, 2019, we
acquire will be based our stock price performancehave acquired all of Mr. Boissel’s free shares upon the exercise of his free share put right in exchange for cash payments from
us totaling €458,149, or approximately $528,000. Neither the
July 2018 announcemententry into of the
transactions contemplated by the TxCell acquisition agreements through the time of purchase of such shares. We have not yet acquired any TxCell free shares fromliquidity agreement with Mr. Boissel
pursuant to such arrangement.The aforementioned TxCell share purchase agreement was not pre-approvednor the exercise of his put right were approved as a related party transaction by our Audit Committee, pursuant to our policy, as Mr. Boissel was not a related person at such time.the time the liquidity agreement was entered into. However, our Audit Committee will provide appropriate reviewreviewed and provided oversight of the implementation of this agreement, and any futurefree share purchases from Mr. Boissel pursuant theretounder his liquidity agreement consistent with applicable Nasdaq rules.
In February 2020, we entered into a global licensing collaboration agreement with Biogen for the research, development and commercialization of gene regulation therapies for the treatment of neurological diseases which became effective in April 2020. Our collaboration aims to leverage our proprietary ZFP technology delivered via AAV to modulate expression of key genes involved in neurological diseases. Concurrently with the execution of the collaboration agreement, we also entered into a stock purchase agreement with BIMA, pursuant to which BIMA purchased 24,420,157 shares of our common stock, or the Biogen Shares, for an aggregate purchase price of $225.0 million. Under the collaboration agreement, Biogen paid us an upfront license fee payment of $125.0 million. We are also eligible to earn research, development, regulatory and commercial milestone payments that could total up to approximately $2.37 billion if Biogen selects all of the targets allowed under the agreement and all the specified milestones set forth in the agreement are achieved, which includes up to $925.0 million in pre-approval milestone payments and up to $1.45 billion in first commercial sale and other sales-based milestone payments. In addition, we are also eligible to receive tiered high single-digit to sub-teen royalties on potential net commercial sales of licensed products arising from the collaboration. These royalty payments are subject to reduction due to patent expiration, entry of biosimilar products to the market and payments made under certain licenses for third-party intellectual property. Under the collaboration agreement, we granted to Biogen an exclusive, royalty bearing and worldwide license, under our relevant patents and know-how, to develop, manufacture and commercialize certain ZFP and/or AAV-based products directed to up to 12 neurological disease gene targets selected by Biogen. Biogen has already selected three of these: ST-501 for tauopathies including Alzheimer’s disease, ST-502 for synucleinopathies including Parkinson’s disease and a third undisclosed neuromuscular disease target. Biogen has exclusive rights to nominate up to nine additional targets over a target selection period of five years. For each gene target selected by Biogen, we perform early research activities, costs for which are shared by the companies, aimed at the development of the combination of proprietary central nervous system delivery vectors and ZFP-TFs targeting therapeutically relevant genes. Biogen then assumes responsibility and costs for the IND-enabling studies, clinical development, related regulatory interactions, and global commercialization. We are primarily responsible for GMP manufacturing activities for the initial clinical studies for the first three products of the collaboration and plan to leverage our in-house manufacturing capacity. Biogen is responsible for GMP manufacturing activities beyond the first clinical study for each of the first three products. Subject to certain exceptions set forth in the collaboration agreement, we are prohibited from developing, manufacturing or commercializing any therapeutic product directed to the targets selected by Biogen. The collaboration agreement continues, on a product-by-product and country-by-country basis, until the expiration of the applicable royalty term. Biogen has the right to terminate the collaboration agreement, in its entirety or on target-by-target basis, for any reason after a specified notice period. Each party has the right to terminate this agreement on account of the other party’s bankruptcy or material, uncured breach. In addition, we may terminate the collaboration agreement if Biogen challenges any
patents licensed by us to Biogen. Pursuant to the terms of the stock purchase agreement, Biogen has agreed not to, without our prior written and subject to specified conditions and exceptions, directly or indirectly acquire shares of our outstanding common stock, seek or propose a tender or exchange offer or merger between the parties, solicit proxies or consents with respect to any matter, or undertake other specified actions related to the potential acquisition of additional equity interests in us. Subject to customary exceptions, such standstill restrictions expire on the earlier of the three-year anniversary of the effectiveness of the Biogen collaboration agreement and the date that Biogen beneficially owns less than 5% of our common stock. The stock purchase agreement also provides that until the first anniversary of the effectiveness of the Biogen collaboration agreement, Biogen must hold and not sell any of the Biogen Shares and from the first anniversary through the second anniversary, Biogen must hold and not sell at least 50% of the Biogen Shares, in addition to being subject to certain volume limitations. The stock purchase agreement further provides that, subject to certain limitations, upon Biogen’s request, we must register for resale any of the Biogen Shares on a registration statement to be filed with the SEC, until such time as all remaining Biogen Shares may be sold pursuant to Rule 144 promulgated under the Securities Act during any 90-day period. In addition, Biogen has agreed that, excluding specified extraordinary matters, it must vote the Biogen Shares in accordance with our recommendation and has granted us an irrevocable proxy with respect to the foregoing. Such voting provisions expire on the earlier of (i) the two-year anniversary of the effectiveness of the Biogen collaboration agreement, (ii) the date that Biogen beneficially owns less than 5% of our common stock and (iii) the date the Biogen collaboration agreement is terminated; provided, however, that in no event shall such expiration date be prior to the one-year anniversary of the effectiveness of the Biogen collaboration agreement.
Indemnification.In addition to the indemnification provisions contained in our Seventh Amended and Restated Certificate of Incorporation, as amended, we have entered into separate indemnification agreements with each of our directors and executive officers containing provisions that may require us to, among other things, indemnify them against certain liabilities that may arise by reason of their status or service as directors.POLICIES AND PROCEDURES
Consistent with the requirement under Nasdaq listing rules, the Audit Committee of the Board of Directors is responsible for reviewing and approving all related party transactions as defined under SEC rules and regulations. While we do not have a formal written policy or procedure for the review, approval or ratification of related party transactions, the Audit Committee must review the material facts of any such transaction and approve that transaction.
To identify related party transactions, each year we submit and require our directors and officers to complete director and officer questionnaires identifying transactions with us in which the director or officer or their family members have a conflict of interest.executive officers. We review the questionnaire for potential related party transactions. In addition, at meetings of the Audit Committee, management may recommend related party transactions to the committee, including the material terms of the proposed transactions, for its consideration. In making its decision to approve or ratify a related party transaction, the Audit Committee will consider all relevant facts and circumstances available to the committee, including factors suchamend these indemnification agreements from time-to-time as the aggregate value of the transaction, whether the terms of the related party transaction are no less favorable than terms generally available in an arms’ length transaction and the benefit of such transaction to us.
61
appropriate.
SECTION 16(a) BENEFICIAL OWNERSHIP REPORTING COMPLIANCE
The members of the Board of Directors, our executive officers and persons who beneficially own more than 10% of our outstanding common stock are subject to the reporting requirements of Section 16 of the Exchange Act, as amended, which require them to file reports with respect to their beneficial ownership of the common stock and their transactions in such common stock. Based upon (i) the copies of Section 16 reports that we received from such persons for their 2018 year transactions in the common stock and their common stock holdings, and (ii) written representation that no other reports were required, we believe that all reporting requirements under Section 16 for such year were met in a timely manner by our directors, executive officers and greater than 10% beneficial owners, except as follows: each of Alexander D. Macrae, Kathy Y. Yi, and Edward R. Conner, three of our named executive officers, and Curt A. Herberts, III, a former executive officer, filed one late Form 4 each of which was filed three days late reporting the annual grants of stock options and RSUs to such executive officers in January 2018 under the Company’s 2018 Plan.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Proxy Statement contains forward-looking statements regarding our current expectations. These forward-looking statements include, without limitation, statements regarding:
the goals and objectives of our compensation programs; our business
strategies, including our goal to become a sustainable, fully-integrated biopharmaceutical company; potential future payments under our global collaborationstrategy and
license agreement with Kite Pharma; the anticipated benefits to us of our acquisition of TxCell; our plans with respect to, and the anticipated benefits of, second-generation ZFNs, including our plans to evaluate second-generation ZFNs in a new clinical trial and expected related determinations and the timing thereof; other planned studies; the therapeutic potential
forof our product candidates;
anticipated plans and timeliness of us and our collaborators for conducting clinical trials and bringing in-house manufacturing facilities online; and other statements that are not historical fact. In some cases, you can identify forward-looking statements by terms such as:
“will,“anticipates,”
“intend,“believes,”
“anticipate,” “believe,” “continue,“continues,” “could,”
“estimate,“estimates,”
“expect,“expects,” “intends,” “may,”
“plan,“plans,”
“seek,“seeks,” “should,” and
“should.“will.” These statements are not guarantees of future performance and are subject to certain risks, uncertainties and assumptions that are difficult to predict. Factors that could cause actual results to differ include, but are not limited to, risks and uncertainties related to:
our dependencethe effects of the ongoing COVID-19 pandemic and the impacts of the pandemic on the
successglobal business environment, healthcare systems and business and operations of Sangamo and our collaborators, including the initiation and operation of clinical trials; the
lengthyconstruction, opening and
operation of in-house manufacturing facilities; the research and development process; and the uncertain
regulatory approval process,timing and unpredictable results of clinical trials, including
the risk thatwhether initial clinical trial data
are subject to differing interpretations and assessments by regulatory authorities; uncertainties related to the initiation, enrollment and completionwill be representative of
final clinical
trials; the possibility of unfavorable new clinicaltrial data and
further analyseswhether final clinical trial data will validate the safety and efficacy of
existing clinical data; whether we will be ableproduct candidates. Actual results may differ from those projected in forward-looking statements due to
effectively deliverrisks and uncertainties that exist in our
ZFNs to produce a beneficial therapeutic effect, including the risks that second-generation ZFNs may not be successfully integrated into our product candidatesoperations and
even if successfully integrated, second-generation ZFNs may not have any advantages over our first-generation ZFNs or otherwise may not produce any beneficial therapeutic effect; our ability to realize the anticipated benefits of our acquisition of TxCell, including the risk that we may not be successful at developing a CAR-Treg therapy that can be used in patients; competitive developments; our reliance on partners and other third-parties to meet their clinical and manufacturing obligations; our ability to maintain strategic partnerships, including our global collaboration and license agreement with Kite Pharma; and the possibility that our compensation programs may not result in improved performance. Further, there can be no assurance that the necessary regulatory approvals for BIVV003,ST-920,SB-525,SB-913,SB-318,SB-FIX or any other product candidates will be obtained or that we and our partners will be able to develop commercially viable product candidates.business environments. These risks and uncertainties are described more fully in
our Annual Report onthe 2020 Form
10-K for the year ended December 31, 2018 10‑K as filed with the
Securities and Exchange Commission.SEC. Forward-looking statements contained in this Proxy Statement are made as of the date of this Proxy Statement, and we undertake no duty to update such information except as required under applicable law.
HOUSEHOLDING OF PROXY MATERIALS
The SEC has adopted rules that permit companies and intermediaries
(such as brokers) to satisfy the delivery requirements for Notices
of Internet Availability of Proxy Materials or other Annual Meetingand proxy materials with respect to two or more stockholders sharing the same address by delivering a single Notice
or a single set of
Internet Availability of Proxy62
Materials or other Annual Meetingproxy materials, as applicable, addressed to those stockholders. This process, which is commonly referred to as “householding,” potentially means extra convenience for stockholders and cost savings for companies.
This year, a number of brokers with account holders who are our stockholders will be “householding” Notices and our proxy materials. A single Notice or a single set of Internet Availability of Proxy Materialsproxy materials, as applicable, will be delivered to multiple stockholders sharing an address unless contrary instructions have been received from the affected stockholders. Once you have received notice from your broker that theyit will be “householding” communications to your address, “householding” will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no longer wish to participate in “householding” and would prefer to receive a separate Notice or set of Internet Availability of Proxy Materials,proxy materials, please notify your broker or us. Direct your written request to Sangamo Therapeutics, Inc., Kathy Y. Yi,Gary H. Loeb, Corporate Secretary, 501 Canal7000 Marina Boulevard, Richmond,Brisbane, California 9480494005 or contact McDavid StilwellAron Feingold at510-970-6000. Stockholders who currently receive multiple copies of the NoticesNotice or sets of Internet Availability of Proxy Materialsproxy materials at their addressesaddress and would like to request “householding” of their communications should contact their brokers.broker. In addition, we will promptly deliver, upon written or oral request to the address or telephone number above, a separate copy of the Noticea
Notice or the full set of proxy materials, as applicable, to a stockholder at a shared address to which a single
copyNotice or set of the
documentsproxy materials, as applicable, was delivered.
OTHER MATTERS
The Board of Directors knows of no other business that will be presented to the Annual Meeting. If any other business is properly brought before the Annual Meeting, or with respect to any adjournment or postponement thereof, it is the intention of the proxy holders to vote on such matters in accordance with their best judgment.
It is important that your shares are represented at the Annual Meeting. We urge you to vote your shares via the Internet, over the telephone or, if you received a paper proxy card or voting instruction form by mail, by marking, dating and signing the proxy card or voting instruction form and mailing it promptly in the return envelope provided.
| | | | | |
| By Order of the Board of Directors, |
| |
| /s/ GARY H. LOEB |
| Gary H. Loeb
Corporate Secretary |
| Brisbane, California | |
| April 2, 2021 | |
Our Annual Report on Form
10-K for the year ended December 31,
20182020 is available without charge upon written request to: Corporate Secretary, Sangamo Therapeutics, Inc.,
501 Canal7000 Marina Boulevard,
Richmond,Brisbane, California
94804.94005. Our Annual Report on Form
10-K is not incorporated into this Proxy Statement and is not considered proxy soliciting material.
|
THE BOARD OF DIRECTORS OF |
SANGAMO THERAPEUTICS, INC. |
Dated: April 25, 2019
63

Appendix A
Sangamo Therapeutics, Inc.
2020 Employee Stock Purchase Plan
Approved by the Compensation Committee of the Board: September 18, 2020
Approved by the Stockholders: May [___], Ch.B.2021
1.General.
(a)Purpose. The Company, by means of the Plan, seeks to retain the services of such Employees, to secure and retain the services of Employees and to provide incentives for such persons to exert maximum efforts for the success of the Company and its Related Corporations. The Plan provides a means by which Eligible Employees of the Company and certain designated Related Corporations may be given an opportunity to purchase shares of Common Stock. The Plan permits the Company to grant a series of Purchase Rights to Eligible Employees under an Employee Stock Purchase Plan. In addition, the Plan permits the Company to grant a series of Purchase Rights to Eligible Employees that do not meet the requirements of an Employee Stock Purchase Plan. Capitalized terms used in the Plan have the meanings set forth in Section 16.
(b)Qualified and Non-Qualified Offerings Permitted. The Plan includes two components: a 423 Component and a Non-423 Component. The Company intends (but makes no undertaking or representation to maintain) the 423 Component to qualify as an Employee Stock Purchase Plan. The provisions of the 423 Component, accordingly, will be construed in a manner that is consistent with the requirements of Section 423 of the Code. Except as otherwise provided in the Plan or determined by the Committee, the Non-423 Component will operate and be administered in the same manner as the 423 Component.
2.Administration.
(a)Administration by Committee. The Committee will administer the Plan pursuant to the delegation of authority to the Committee as set forth in the Committee’s charter, unless otherwise determined by the Board. The Board retains concurrent authority to administer the Plan. To the extent the Board administers the Plan, references herein to the Committee shall be deemed to refer to the Board except where context dictates otherwise.
(b)Powers of Committee. The Committee will have the power, subject to, and within the limitations of, the express provisions of the Plan:
(i)To determine how and when Purchase Rights will be granted and the provisions of each Offering (which need not be identical).
(ii)To designate from time to time (A) which Related Corporations of the Company will be eligible to participate in the Plan, (B) whether such Related Corporations will participate in the 423 Component or the Non-423 Component, and (C) to the extent that the Company makes separate Offerings under the 423 Component, in which Offering the Related Corporations in the 423 Component will participate.
(iii)To construe and interpret the Plan and Purchase Rights, and to establish, amend and revoke rules and regulations for its administration. The Committee, in the exercise of this power, may correct any defect, omission or inconsistency in the Plan, in a manner and to the extent it deems necessary or expedient to make the Plan fully effective.
(iv)To settle all controversies regarding the Plan and Purchase Rights granted under the Plan.
(v)To suspend or terminate the Plan at any time as provided in Section 12.
(vi)To amend the Plan at any time as provided in Section 12.
(vii)Generally, to exercise such powers and to perform such acts as it deems necessary or expedient to promote the best interests of the Company and its Related Corporations and to carry out the intent that the Plan be treated as an Employee Stock Purchase Plan with respect to the 423 Component.
(viii)To adopt such rules, procedures and sub-plans as are necessary or appropriate to permit or facilitate participation in the Plan by Employees who are foreign nationals or employed or located outside the United States. Without limiting the generality of, and consistent with, the foregoing, the Committee specifically is authorized to adopt rules, procedures, and sub-plans regarding, without limitation, eligibility to participate in the Plan, the definition of eligible “earnings,” handling and making of Contributions, establishment of bank or trust accounts to hold Contributions, payment of interest, conversion of local currency, obligations to pay payroll tax, determination of beneficiary designation requirements, withholding procedures and handling of share issuances, any of which may
vary according to applicable requirements, and which, if applicable to a Related Corporation designated for participation in the Non-423 Component, do not have to comply with the requirements of Section 423 of the Code.
(c)Delegation of Powers. The Committee will have the power to delegate to a subcommittee any of the administrative powers the Committee is authorized to exercise (and references to the Committee in this Plan and in any applicable Offering Document will thereafter be to such subcommittee, as applicable, except where context dictates otherwise), Ph.D. 07 - Joseph S. Zakrzewski 02 - Robert F. Carey 05 - Saira Ramasastry 03 - Stephen G. Dilly, M.B.B.S.subject, however, to such resolutions, not inconsistent with the provisions of the Plan, as may be adopted from time to time. The Committee retains the authority to concurrently administer the Plan with any subcommittee. The Committee will have the final power to determine all questions of policy and expediency that may arise in the administration of the Plan.
(d)Effect of Committee’s Decisions. All determinations, interpretations and constructions made by the Committee in good faith will not be subject to review by any person and will be final, binding and conclusive on all persons.
3.Common Stock Subject to the Plan.
(a)Number of Shares Available. Subject to the provisions of Section 11(a) relating to Capitalization Adjustments, the maximum number of shares of Common Stock that may be issued under the Plan will not exceed 5,000,000 shares of Common Stock. For the avoidance of doubt, up to the maximum number of Common Stock reserved under this Section 3(a) may be used to satisfy purchases of Common Stock under the 423 Component and any remaining portion of such maximum number of shares may be used to satisfy purchases of Common Stock under the Non-423 Component.
(b)Share Recycling. If any Purchase Right granted under the Plan terminates without having been exercised in full, the Common Stock not purchased under such Purchase Right will again become available for issuance under the Plan.
(c)Source of Shares. The shares purchasable under the Plan will be authorized but unissued or reacquired Common Stock, including Common Stock repurchased by the Company on the open market.
4.Grant of Purchase Rights; Offering.
(a)Offerings. The Committee may from time to time grant or provide for the grant of Purchase Rights to Eligible Employees under an Offering (consisting of one or more Purchase Periods) on an Offering Date or Offering Dates selected by the Committee. Each Offering will be in such form and will contain such terms and conditions as the Committee will deem appropriate, and, with respect to the 423 Component, will comply with the requirement of Section 423(b)(5) of the Code that all Employees granted Purchase Rights will have the same rights and privileges. The terms and conditions of an Offering shall be incorporated by reference into the Plan and treated as part of the Plan. The provisions of separate Offerings need not be identical, but each Offering will include (through incorporation of the provisions of this Plan by reference in the document comprising the Offering or otherwise) the period during which the Offering will be effective, which period will not exceed 27 months beginning with the Offering Date, and the substance of the provisions contained in Sections 5 through 8, inclusive.
(b)Multiple Purchase Rights. If a Participant has more than one Purchase Right outstanding under the Plan, unless he or she otherwise indicates in forms delivered to the Company: (i) each form will apply to all of his or her Purchase Rights under the Plan, and (ii) a Purchase Right with a lower exercise price (or an earlier-granted Purchase Right, if different Purchase Rights have identical exercise prices) will be exercised to the fullest possible extent before a Purchase Right with a higher exercise price (or a later-granted Purchase Right if different Purchase Rights have identical exercise prices) will be exercised.
(c)Restart Provision Permitted. The Committee will have the discretion to structure an Offering so that if the Fair Market Value of a share of Common Stock on the first Trading Day of a new Purchase Period within that Offering is less than or equal to the Fair Market Value of a share of Common Stock on the Offering Date for that Offering, then (i) that Offering will terminate immediately as of that first Trading Day, and (ii) the Participants in such terminated Offering will be automatically enrolled in a new Offering beginning on the first Trading Day of such new Purchase Period.
5.Eligibility.
(a)General. Purchase Rights may be granted only to Employees of the Company or, as the Committee may designate in accordance with Section 2(b), Ph.D. 06 - Karen L. Smith, M.D.to Employees of a Related Corporation. Except as provided in Section 5(b) or as required by Applicable Law, an Employee will not be eligible to be granted Purchase Rights unless, on the Offering Date, the Employee has been in the employ of the Company or the Related Corporation, as the case may be, for such continuous period preceding such Offering Date as the Committee may require, but in no event will the
required period of continuous employment be equal to or greater than two years. In addition, the Committee (unless prohibited by law) may provide that no Employee will be eligible to be granted Purchase Rights under the Plan unless, on the Offering Date, such Employee’s customary employment with the Company or the Related Corporation is more than 20 hours per week and more than five months per calendar year or such other criteria as the Committee may determine consistent with Section 423 of the Code with respect to the 423 Component.
(b)Grant of Purchase Rights. The Committee may provide that each person who, during the course of an Offering, first becomes an Eligible Employee will, on a date or dates specified in the Offering which coincides with the day on which such person becomes an Eligible Employee or which occurs thereafter, receive a Purchase Right under that Offering, which Purchase Right will thereafter be deemed to be a part of that Offering. Such Purchase Right will have the same characteristics as any Purchase Rights originally granted under that Offering, as described herein, except that:
(i)the date on which such Purchase Right is granted will be the Offering Date of such Purchase Right for all purposes, including determination of the exercise price of such Purchase Right;
(ii)the period of the Offering with respect to such Purchase Right will begin on its Offering Date and end coincident with the end of such Offering; and
(iii)the Committee may provide that if such person first becomes an Eligible Employee within a specified period of time before the end of the Offering, he or she will not receive any Purchase Right under that Offering.
(c)5% Stockholders. No Employee will be eligible for the grant of any Purchase Rights if, immediately after any such Purchase Rights are granted, such Employee owns shares possessing five percent or more of the total combined voting power or value of all classes of stock of the Company or of any Related Corporation. For purposes of this Section 5(c), Ph.D., M.B.A., L.L.M. For Withhold For Withhold For Withhold 7 2 C V Usingthe rules of Section 424(d) of the Code will apply in determining the stock ownership of any Employee, and stock which such Employee may purchase under all outstanding Purchase Rights and options will be treated as stock owned by such Employee.
(d)$25,000 Limit. As specified by Section 423(b)(8) of the Code, an Eligible Employee may be granted Purchase Rights only if such Purchase Rights, together with any other rights granted under all Employee Stock Purchase Plans of the Company and any Related Corporations, do not permit such Eligible Employee’s rights to purchase stock of the Company or any Related Corporation to accrue at a black ink pen, mark your votesrate which, when aggregated, exceeds U.S. $25,000 of Fair Market Value of such stock (determined at the time such rights are granted, and which, with respect to the Plan, will be determined as of their respective Offering Dates) for each calendar year in which such rights are outstanding at any time.
(e)Officers and Highly Compensated Employees. Officers of the Company and any designated Related Corporation, if they are otherwise Eligible Employees, will be eligible to participate in Offerings under the Plan. Notwithstanding the foregoing, the Committee may (unless prohibited by law) provide in an Offering that Employees who are highly compensated Employees within the meaning of Section 423(b)(4)(D) of the Code will not be eligible to participate.
(f)Non 423 Component Offerings. Notwithstanding anything in this Section 5 to the contrary, in the case of an Offering under the Non-423 Component, an Eligible Employee (or group of Eligible Employees) may be excluded from participation in the Plan or an Offering if the Committee has determined, in its sole discretion, that participation of such Eligible Employee(s) is not advisable or practical for any reason.
6.Purchase Rights; Purchase Price.
(a)Grant and Maximum Contribution Rate. On each Offering Date, each Eligible Employee, pursuant to an Offering made under the Plan, will be granted a Purchase Right to purchase up to that number of shares of Common Stock purchasable either with a percentage of such Employee’s earnings or with a maximum dollar amount (as defined by the Committee in each Offering) during the period that begins on the Offering Date (or such later date as the Committee determines for a particular Offering) and ends on the date stated in the Offering, which date will be no later than the end of the Offering.
(b)Purchase Dates. The Committee will establish one or more Purchase Dates during an Offering on which Purchase Rights granted for that Offering will be exercised and Common Stock will be purchased in accordance with such Offering.
(c)Purchase Limits. In connection with each Offering made under the Plan, the Committee may specify (i) a maximum number of shares of Common Stock that may be purchased by any Participant on any Purchase Date during such Offering, (ii) a maximum aggregate number of shares of Common Stock that may be purchased by all
Participants pursuant to such Offering and/or (iii) a maximum aggregate number of shares of Common Stock that may be purchased by all Participants on any Purchase Date under the Offering. If the aggregate number of shares of Common Stock issuable upon exercise of Purchase Rights granted under the Offering would exceed any such maximum aggregate number, then, in the absence of any Committee action otherwise, a pro rata (based on each Participant’s accumulated Contributions) allocation of the shares of Common Stock (rounded down to the nearest whole share) available will be made in as nearly a uniform manner as will be practicable and equitable.
(d)Purchase Price. The purchase price of shares of Common Stock acquired pursuant to Purchase Rights will be not less than the lesser of:
(i)an amount equal to 85% of the Fair Market Value of the Common Stock on the Offering Date; or
(ii)an amount equal to 85% of the Fair Market Value of the Common Stock on the applicable Purchase Date.
7.Participation; Withdrawal; Termination.
(a)Enrollment and Contributions. An Eligible Employee may elect to participate in an Offering and authorize payroll deductions as the means of making Contributions by completing and delivering to the Company, within the time specified in the Offering, an enrollment form provided by the Company. The enrollment form will specify the amount of Contributions not to exceed the maximum amount specified by the Committee. Each Participant’s Contributions will be credited to a bookkeeping account for such Participant under the Plan and will be deposited with the general funds of the Company except where Applicable Law requires that Contributions be deposited with a third party. If permitted in the Offering, a Participant may begin such Contributions with the first practicable payroll occurring on or after the Offering Date (or, in the case of a payroll date that occurs after the end of the prior Offering but before the Offering Date of the next new Offering, Contributions from such payroll will be included in the new Offering). If permitted in the Offering or required by Applicable Law, a Participant may thereafter reduce (including to zero) or increase his or her Contributions. If specifically provided in the Offering, in addition to or instead of making Contributions by payroll deductions, a Participant may make Contributions through the payment by cash or check prior to a Purchase Date or such other payment method specified by the Committee for such Offering.
(b)Withdrawals. During an Offering, a Participant may cease making Contributions and withdraw from the Offering by delivering to the Company a withdrawal form provided by the Company. The Company may impose a deadline before a Purchase Date for withdrawing. Upon such withdrawal, such Participant’s Purchase Right in that Offering will immediately terminate and the Company will distribute as soon as practicable to such Participant all of his or her accumulated but unused Contributions and such Participant’s Purchase Right in that Offering shall thereupon terminate. A Participant’s withdrawal from that Offering will have no effect upon his or her eligibility to participate in any other Offerings under the Plan, but such Participant will be required to deliver a new enrollment form to participate in subsequent Offerings.
(c)Termination of Eligibility. Unless otherwise required by Applicable Law, Purchase Rights granted pursuant to any Offering under the Plan will terminate immediately if the Participant either (i) is no longer an Employee for any reason or for no reason (subject to any post-employment participation period required by law) or (ii) is otherwise no longer eligible to participate. The Company will distribute to such individual as soon as practicable all of his or her accumulated but unused Contributions.
(d)Employee Transfers. Unless otherwise determined by the Committee, a Participant whose employment transfers or whose employment terminates with an Ximmediate rehire (with no break in service) by or between the Company and a Related Corporation that has been designated for participation in the Plan will not be treated as shownhaving terminated employment for purposes of participating in this example. Please do not write outside the designated areas. 03219D + + Proposals — The Board of Directors recommendPlan or an Offering; however, if a vote FOR allParticipant transfers from an Offering under the nominees listed A and FOR Proposals 2 – 3. 2. To approve, on423 Component to an advisory basis,Offering under the compensationNon-423 Component, the exercise of the Company’s named executive officers,Participant’s Purchase Right will be qualified under the 423 Component only to the extent such exercise complies with Section 423 of the Code. If a Participant transfers from an Offering under the Non-423 Component to an Offering under the 423 Component, the exercise of the Purchase Right will remain non-qualified under the Non-423 Component. The Committee may establish different and additional rules governing transfers between separate Offerings within the 423 Component and between Offerings under the 423 Component and Offerings under the Non-423 Component.
(e)No Transfer of Purchase Rights. During a Participant’s lifetime, Purchase Rights will be exercisable only by such Participant. Purchase Rights are not transferable by a Participant, except by will, by the laws of descent and distribution, or, if permitted by the Company, by a beneficiary designation as described in Section 10.
(f)No Interest. Unless otherwise specified in the Proxy Statement. 3. To ratifyOffering or required by Applicable Law, the appointmentCompany will have no obligation to pay interest on Contributions.
8.Exercise of Purchase Rights.
(a)Accumulated Contributions. On each Purchase Date, each Participant’s accumulated Contributions will be applied to the purchase of shares of Common Stock (rounded down to the nearest whole share), up to the maximum number of shares of Common Stock permitted by the Plan and the applicable Offering, at the purchase price specified in the Offering. No fractional shares will be issued unless specifically provided for in the Offering.
(b)Remaining Contributions. Unless otherwise provided in the Offering, if any amount of accumulated Contributions remains in a Participant’s account after the purchase of shares of Common Stock and such remaining amount is less than the amount required to purchase one share of Common Stock on the final Purchase Date of an Offering, then such remaining amount will be held in such Participant’s account for the year ending December 31, 2019. 1.purchase of Common Stock under the next Offering under the Plan, unless such Participant withdraws from or is not eligible to participate in such next Offering, in which case such amount will be distributed to such Participant after the final Purchase Date without interest (unless the payment of interest is otherwise required by Applicable Law). If the amount of Contributions remaining in a Participant’s account after the purchase of Common Stock is at least equal to the amount required to purchase one (1) whole share of Common Stock on the final Purchase Date of an Offering, then such remaining amount will be refunded in full to such Participant after the final Purchase Date of such Offering without interest (unless the payment of interest is otherwise required by Applicable Law).
(c)Limitations on Exercise. No Purchase Rights may be exercised to any extent unless the shares of Common Stock to be issued upon such exercise under the Plan are covered by an effective registration statement pursuant to the Securities Act and the Plan is in material compliance with all applicable U.S. federal and state, foreign and other securities, exchange control and other laws applicable to the Plan. If on a Purchase Date the shares of Common Stock are not so registered or the Plan is not in such compliance, no Purchase Rights will be exercised on such Purchase Date, and the Purchase Date will be delayed until the shares of Common Stock are subject to such an effective registration statement and the Plan is in material compliance, except that the Purchase Date will in no event be more than 27 months from the Offering Date. If, on the Purchase Date, as delayed to the maximum extent permissible, the shares of Common Stock are not registered and the Plan is not in material compliance with all Applicable Laws, as determined by the Company in its sole discretion, no Purchase Rights will be exercised and all accumulated but unused Contributions will be distributed as soon as practicable to the Participants without interest (unless the payment of interest is otherwise required by Applicable Law).
9.Authorizations.
(a)U.S. Participants. With respect to U.S. Participants the Company will seek to obtain from each Governing Entity such authority as may be required to grant Purchase Rights and issue and sell Common Stock thereunder to such Participants unless the Company determines, in its sole discretion, that doing so is not practical or would cause the Company to incur costs that are unreasonable. If, after commercially reasonable efforts, the Company is unable to obtain the authority that counsel for the Company deems necessary for the grant of Purchase Rights or the lawful issuance and sale of Common Stock under the Plan to U.S. Participants, and at a commercially reasonable cost, the Company will be relieved from any liability for failure to grant Purchase Rights and/or to issue and sell Common Stock upon exercise of such Purchase Rights to such Participants.
(b)Non-U.S. Participants. With respect to Non-U.S. Participants the Company may, but is not obligated to, seek to obtain from each Governing Entity such authority as may be required to grant Purchase Rights and issue and sell Common Stock thereunder to such Participants. If the Company does not obtain the authority that counsel for the Company deems necessary for the grant of Purchase Rights or the lawful issuance and sale of Common Stock under the Plan to Non-U.S. Participants, the Company will be relieved from any liability for failure to grant Purchase Rights and/or to issue and sell Common Stock upon exercise of such Purchase Rights to such Participants.
10.Participant Beneficiaries.
(a)Beneficiary Designation. The Company may, but is not obligated to, permit a Participant to submit a form designating a beneficiary who will receive any shares or Common Stock and/or Contributions from the Participant’s account under the Plan if the Participant dies before such shares and/or Contributions are delivered to the Participant. The Company may, but is not obligated to, permit the Participant to change such designation of beneficiary. Any such designation and/or change must be on a form approved by the Company.
(b)Death of Participant. If a Participant dies, and in the absence of a valid beneficiary designation, the Company will deliver any shares of Common Stock and/or Contributions to the executor or administrator of the estate of the Participant. If no executor or administrator has been appointed (to the knowledge of the Company), the Company, in its sole discretion, may deliver such shares of Common Stock and/or Contributions without interest (unless the payment of interest is otherwise required by Applicable Law), to the Participant’s spouse, dependents or
relatives, or if no spouse, dependent or relative is known to the Company, then to such other person as the Company may designate.
11.Adjustments upon Changes in Common Stock; Corporate Transactions.
(a)Capitalization Adjustment. In the event of a Capitalization Adjustment, the Committee will appropriately and proportionately adjust: (i) the class(es) and maximum number of securities subject to the Plan pursuant to Section 3(a), (ii) the class(es) and number of securities subject to, and the purchase price applicable to outstanding Offerings and Purchase Rights, and (iii) the class(es) and number of securities that are the subject of the individual and any aggregate purchase limits under each ongoing Offering. The Committee will make these adjustments, and its determination will be final, binding and conclusive.
(b)Corporate Transaction. In the event of a Corporate Transaction, then: (i) any surviving corporation or acquiring corporation (or the surviving or acquiring corporation’s parent company) may assume or continue outstanding Purchase Rights or may substitute similar rights (including a right to acquire the same consideration paid to the stockholders in the Corporate Transaction) for outstanding Purchase Rights, or (ii) if any surviving or acquiring corporation (or its parent company)does not assume or continue such Purchase Rights or does not substitute similar rights for such Purchase Rights, then the Participants’ accumulated Contributions will be used to purchase shares of Common Stock (rounded down to the nearest whole share) within ten business days prior to the Corporate Transaction under the outstanding Purchase Rights, and the Purchase Rights will terminate immediately after such purchase.
12.Amendment, Termination or Suspension of the Plan.
(a)Plan Amendment. The Committee may amend the Plan at any time in any respect the Committee deems necessary or advisable. However, except as provided in Section 11(a) relating to Capitalization Adjustments, stockholder approval will be required for any amendment of the Plan for which stockholder approval is required by Applicable Law.
(b)Suspension or Termination. The Committee may suspend or terminate the Plan at any time. No Purchase Rights may be granted under the Plan while the Plan is suspended or after it is terminated.
(c)No Material Impairment of Rights. Any benefits, privileges, entitlements and obligations under any outstanding Purchase Rights granted before an amendment, suspension or termination of the Plan will not be materially impaired by any such amendment, suspension or termination except (i) with the consent of the person to whom such Purchase Rights were granted, (ii) as necessary to comply with any laws, listing requirements, or governmental regulations (including, without limitation, the provisions of Section 423 of the Code and the regulations and other interpretive guidance issued thereunder relating to Employee Stock Purchase Plans) including without limitation any such regulations or other guidance that may be issued or amended after the date the Plan is adopted by the Committee, or (iii) as necessary to obtain or maintain favorable tax, listing, or regulatory treatment. To elect seven (7) directorsbe clear, the Committee may amend outstanding Purchase Rights without a Participant’s consent if such amendment is necessary to serveensure that the Purchase Right and/or the Plan complies with the requirements of Section 423 of the Code with respect to the 423 Component or with respect to other Applicable Laws.
(d)Corrections and Administrative Procedures. Notwithstanding anything in the Plan or any Offering Document to the contrary, the Committee will be entitled to: (i) establish the exchange ratio applicable to amounts withheld in a currency other than U.S. dollars; (ii) permit Contributions in excess of the amount designated by a Participant in order to adjust for mistakes in the Company’s processing of properly completed Contribution elections; (iii) establish reasonable waiting and adjustment periods and/or accounting and crediting procedures to ensure that amounts applied toward the purchase of Common Stock for each Participant properly correspond with amounts withheld from the Participant’s Contributions; (iv) amend any outstanding Purchase Rights or clarify any ambiguities regarding the terms of any Offering to enable the Purchase Rights to qualify under and/or comply with Section 423 of the Code with respect to the 423 Component; and (v) establish other limitations or procedures as the Committee determines in its sole discretion advisable that are consistent with the Plan. The actions of the Committee pursuant to this paragraph will not be considered to alter or impair any Purchase Rights granted under an Offering as they are part of the initial terms of each Offering and the Purchase Rights granted under each Offering.
13.Tax Qualification; Tax Withholding.
(a)No Guaranteed Tax Treatment. Although the Company may endeavor to (i) qualify a Purchase Right for special tax treatment under the laws of the United States or jurisdictions outside of the United States or (ii) avoid adverse tax treatment, the Company makes no representation to that effect and expressly disavows any covenant to maintain special or to avoid unfavorable tax treatment, notwithstanding anything to the contrary in this Plan. The Company will be unconstrained in its corporate activities without regard to the potential negative tax impact on Participants.
(b)Withholding. Each Participant will make arrangements, satisfactory to the Company and any applicable Related Corporation, to enable the Company or the Related Corporation to fulfill any withholding obligation for Tax-Related Items. Without limitation to the foregoing, the amount necessary to satisfy such withholding obligation may be withheld as determined in the Company's sole discretion to the extent permitted by Applicable Law: (i) from the Participant’s salary or any other cash payment due to the Participant from the Company or a Related Corporation, (ii) from the proceeds of the sale of Common Stock acquired under the Plan, either through a voluntary sale or mandatory sale arranged by the Company, or (ii) any other method approved by the Committee.
14.Effective Date of Plan. The Plan will become effective upon the date it is adopted by the Committee, provided that no Purchase Rights will be exercised unless and until stockholder approval is received within 12 months after such date. Additional stockholder approval must be received prior to the exercise of Purchase Rights (if and to the extent required under Section 12(a) above) if the Plan is materially amended by the Committee.
15.Miscellaneous Provisions.
(a)Electronic Delivery. Any reference herein to a “written” agreement, form, or document will include any agreement, form, or document delivered electronically, filed publicly at www.sec.gov (or any successor website thereto), or posted on the Company’s intranet (or other shared electronic medium controlled by the Company to which the Participant has access). By participating in the Plan, the Participant consents to receive documents by electronic delivery and to participate in the Plan through any online electronic system established and maintained by the Company or another third party selected by the Company. The form of delivery of any Common Stock (e.g., a stock certificate or electronic entry evidencing such shares) shall be determined by the Company.
(b)Use of Proceeds. Proceeds from the sale of shares of Common Stock pursuant to Purchase Rights will constitute general funds of the Company.
(c)Stockholder Rights. A Participant will not be deemed to be the holder of, or to have any of the rights of a holder with respect to, shares of Common Stock subject to Purchase Rights unless and until the Participant’s shares of Common Stock acquired upon exercise of Purchase Rights are recorded in the books of the Company (or its transfer agent).
(d)No Employment or Other Service Rights. The Plan and Offering do not constitute an employment contract. Nothing in the Plan or in the Offering will in any way alter the at-will nature of a Participant’s employment, if applicable, or be deemed to create in any way whatsoever any obligation on the part of any Participant to continue in the employ of the Company or a Related Corporation, or on the part of the Company or a Related Corporation to continue the employment of a Participant.
(e)Choice of Law. The provisions of the Plan will be governed by the laws of the State of Delaware without resort to that state’s conflicts of laws rules.
(f)Severability. If any particular provision of the Plan is found to be invalid or otherwise unenforceable, such provision will not affect the other provisions of the Plan, but the Plan will be construed in all respects as if such invalid provision were omitted.
(g)Interpretation. If any provision of the Plan does not comply with Applicable Law, such provision shall be construed in such a manner as to comply with Applicable Law.
16.Definitions.
As used in the Plan, the following definitions will apply to the capitalized terms indicated below:
(a)“423 Component” means the part of the Plan, which excludes the Non-423 Component, pursuant to which Purchase Rights that satisfy the requirements for an Employee Stock Purchase Plan may be granted to Eligible Employees.
(b)“Applicable Law” means shall mean any applicable securities, federal, state, foreign, material local or municipal or other law, statute, constitution, principle of common law, resolution, ordinance, code, edict, decree, rule, listing rule, regulation, judicial decision, ruling or requirement issued, enacted, adopted, promulgated, implemented or otherwise put into effect by or under the authority of any Governmental Body (or under the authority of the Exchange or the Financial Industry Regulatory Authority).
(c)“Board” means the Board of Directors untilof the next Annual MeetingCompany.
(d)“Capitalization Adjustment” means any change that is made in, or other events that occur with respect to, the Common Stock subject to the Plan or subject to any Purchase Right after the date the Plan is adopted by the Committee without the receipt of Stockholderconsideration by the Company through merger, consolidation, reorganization,
recapitalization, reincorporation, stock dividend, dividend in property other than cash, large nonrecurring cash dividend, stock split, liquidating dividend, combination of shares, exchange of shares, change in corporate structure or other similar equity restructuring transaction, as that term is used in Financial Accounting Standards Board Accounting Standards Codification Topic 718 (or any successor thereto). Notwithstanding the foregoing, the conversion of any convertible securities of the Company will not be treated as a Capitalization Adjustment.
(e)“Code” means the U.S. Internal Revenue Code of 1986, as amended, including any applicable regulations and guidance thereunder.
(f)“Committee” means the Compensation Committee of the Board.
(g)“Common Stock” means the Company’s common stock.
(h)“Company” means Sangamo Therapeutics, Inc., a Delaware corporation.
(i)“Contributions” means the payroll deductions and other additional payments specifically provided for in the Offering that a Participant contributes to fund the exercise of a Purchase Right. A Participant may make additional payments into his or her account if specifically provided for in the Offering, and then only if the Participant has not already had the maximum permitted amount withheld during the Offering through payroll deductions.
(j)“Corporate Transaction” means the consummation, in a single transaction or in a series of related transactions, of any one or more of the following events:
(i)a sale or other disposition of all or substantially all, as determined by the Committee in its sole discretion, of the consolidated assets of the Company and its subsidiaries;
(ii)a sale or other disposition of more than 50% of the outstanding securities of the Company;
(iii)a merger, consolidation or similar transaction following which the Company is not the surviving corporation; or
(iv)a merger, consolidation or similar transaction following which the Company is the surviving corporation but the shares of Common Stock outstanding immediately preceding the merger, consolidation or similar transaction are converted or exchanged by virtue of the merger, consolidation or similar transaction into other property, whether in the form of securities, cash or otherwise.
(k)“Director” means a member of the Board.
(l)“Eligible Employee” means an Employee who meets the requirements set forth in the document(s) governing the Offering for eligibility to participate in the Offering, provided that such Employee also meets the requirements for eligibility to participate set forth in the Plan.
(m)“Employee” means any person, including an Officer or Director, who is “employed” for purposes of Section 423(b)(4) of the Code by the Company or a Related Corporation. However, service solely as a Director, or payment of a fee for such services, will not cause a Director to be held in 2020 or until their successors are duly elected and qualified; Nominees: For Against Abstain NOTE: PLEASE SIGN EXACTLY AS NAME APPEARS HEREON. JOINT OWNERS SHOULD EACH SIGN. WHEN SIGNING AS ATTORNEY, EXECUTOR, ADMINISTRATOR, TRUSTEE OR GUARDIAN, GIVE FULL NAME AND TITLE AS SUCH. Date (mm/dd/yyyy) — Please print date below. Signature 1 — Please keep signatureconsidered an “Employee” for purposes of the Plan.
(n)“Employee Stock Purchase Plan” means a plan that grants Purchase Rights intended to be options issued under an “employee stock purchase plan,” within the box. Signature 2 — Please keep signaturemeaning of Section 423(b) of the Code.
(o)“Exchange” means the stock exchange or established market on which the Common Stock is listed, including but not limited to the New York Stock Exchange, the Nasdaq Stock Market, or any successors thereto.
(p)“Exchange Act” means the U.S. Securities Exchange Act of 1934, as amended and the rules and regulations promulgated thereunder.
(q)“Fair Market Value” means, as of any date, the value of the Common Stock determined as follows:
(i)If the Common Stock is listed on any established stock exchange or traded on any established market, the Fair Market Value of a share of Common Stock will be, unless otherwise determined by the Committee, the closing sales price for such share of Common Stock as quoted on such exchange or market (or the exchange or market with the greatest volume of trading in the Common Stock) on the date of determination, as reported in such source as the Committee deems reliable. Unless otherwise provided by the Committee, if there is no closing sales price for the Common Stock on the date of determination, then the Fair Market Value will be the closing sales price on the last preceding date for which such quotation exists.
(ii)In the absence of such markets for the Common Stock, the Fair Market Value will be determined by the Committee in good faith in compliance with Applicable Laws and in a manner that complies with Sections 409A of the Code.
(r)“Governmental Body” means any: (a) nation, state, commonwealth, province, territory, county, municipality, district or other jurisdiction of any nature; (b) federal, state, local, municipal, foreign or other government; (c) governmental or regulatory body, or quasi-governmental body of any nature (including any governmental division, department, administrative agency or bureau, commission, authority, instrumentality, official, ministry, fund, foundation, center, organization, unit, body or entity and any court or other tribunal, and for the avoidance of doubt, any tax authority) or other body exercising similar powers or authority; or (d) self-regulatory organization (including the Exchange and the Financial Industry Regulatory Authority).
(s)“Governing Entity” means each U.S. federal or state, foreign or other regulatory commission or agency having jurisdiction over the Plan.
(t)“Non-423 Component” means the part of the Plan, which excludes the 423 Component, pursuant to which Purchase Rights that are not intended to satisfy the requirements for an Employee Stock Purchase Plan may be granted to Eligible Employees.
(u)“Non-U.S. Participants” means Participants employed by any Related Corporation that is not incorporated or organized in the United States.
(v)“Offering” means the grant to Eligible Employees of Purchase Rights, with the exercise of those Purchase Rights automatically occurring at the end of one or more Purchase Periods. The terms and conditions of an Offering will generally be set forth in the Offering Document.
(w)“Offering Date” means a date selected by the Committee for an Offering to commence.
(x)Offering Document” the document that sets forth the terms and conditions of an Offering that has been approved by the Committee for that Offering.
(y)“Officer”meansa person who is an officer of the Company or a Related Corporation within the box. B Authorized Signatures — This section must be completed for your vote to count. Please date and sign below. IF VOTING BY MAIL, SIGN, DETACH AND RETURN THE BOTTOM PORTION IN THE ENCLOSED ENVELOPE.q 2019 Annual Meeting Proxy Card For Against Abstain 000004 MR A SAMPLE DESIGNATION (IF ANY) ADD 1 ADD 2 ADD 3 ADD 4 ADD 5 ADD 6 ENDORSEMENT_LINE SACKPACK 1234 5678 9012 345 4 1 6 1 9 2 MR A SAMPLE (THIS AREA IS SET UP TO ACCOMMODATE 140 CHARACTERS) MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND MR A SAMPLE AND C 1234567890 J N T C123456789 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext 000000000.000000 ext If no electronic voting, delete QR code and control # You may vote online or by phone insteadmeaning of mailing this card. Online Go to www.envisionreports.com/SGMO or scan the QR code — login details are located in the shaded bar below. Save paper, time and money! Sign up for electronic delivery at www.envisionreports.com/SGMO Phone Call toll free 1-800-652-VOTE (8683) within the USA, US territories and Canada Votes submitted electronically must be received by 1:00am, EDT, on June 10, 2019. Your vote matters – here’s how to vote!

Small steps make an impact. Help the environment by consenting to receive electronic delivery, sign up at www.envisionreports.com/SGMO PROXY SOLICITED BY THE BOARD OF DIRECTORS FOR THE ANNUAL MEETING OF STOCKHOLDERS, JUNE 10, 2019 The undersigned hereby appoints Alexander D. Macrae, M.B., Ch.B., Ph.D. and Kathy Y. Yi and each of them, as proxiesSection 16 of the undersigned, with full power of substitution, to vote all shares ofExchange Act.
(z)“Participant” means an Eligible Employee who holds an outstanding Purchase Right.
(aa) “Plan” means this Sangamo Therapeutics, Inc. 2020 Employee Stock Purchase Plan, as amended from time to time, including both the 423 Component and the Non-423 Component.
(bb) “Purchase Date” means one or more dates during an Offering selected by the Committee on which Purchase Rights will be exercised and on which purchases of shares of Common Stock will be carried out in accordance with such Offering.
(cc) “Purchase Period” means a period of time specified within an Offering, generally beginning on the Offering Date or on the first Trading Day following a Purchase Date, and ending on a Purchase Date. An Offering may consist of one or more Purchase Periods.
(dd) “Purchase Right” means an option to purchase shares of Common Stock granted pursuant to the Plan.
(ee) “Related Corporation” means any “parent corporation” or “subsidiary corporation” of the Company whether now or subsequently established, as those terms are defined in Sections 424(e) and (f), respectively, of the Code.
(ff) “Securities Act” means the U.S. Securities Act of 1933, as amended.
(gg) “Tax-Related Items” means any income tax, social insurance, payroll tax, fringe benefit tax, payment on account or other tax-related items arising out of or in relation to a Participant’s participation in the Plan, including, but not limited to, the exercise of a Purchase Right and the receipt of shares of Common Stock or the sale or other disposition of shares of Common Stock acquired under the Plan.
(hh) “Trading Day”means any day on which the undersignedExchange is entitled to vote on all matters which may properly come beforeopen for trading.
(ii) “U.S. Participants” means Participants employed by the 2019 Annual MeetingCompany or any Related Corporation that is incorporated or organized in the United States.
Table of Sangamo Therapeutics, Inc., to be held at 7000 Marina Blvd, Brisbane, CA 94005 on June 10, 2019 at 9:00 a.m. PDT or at any postponement or adjournment thereof. THE SHARES REPRESENTED BY THIS PROXY CARD WILL BE VOTED AS SPECIFIED ON THE REVERSE SIDE, BUT IF NO SPECIFICATION IS MADE THEY WILL BE VOTED FOR ALL NOMINEES, FOR PROPOSAL 2 AND FOR PROPOSAL 3 AND AT THE DISCRETION OF THE PROXY ON ANY OTHER MATTER THAT MAY PROPERLY COME BEFORE THE MEETING. (SEE PROXY STATEMENT FOR DISCUSSION OF ITEMS) PLEASE SIGN, DATE AND RETURN PROMPTLY IN THE ACCOMPANYING ENVELOPE. CONTINUED AND TO BE SIGNED ON REVERSE SIDE Proxy — Sangamo Therapeutics, Inc. IF VOTING BY MAIL, SIGN, DETACH AND RETURN THE BOTTOM PORTION IN THE ENCLOSED ENVELOPE. C Non-Voting Items + + Meeting Attendance Mark box to the right if you plan to attend the Annual Meeting. Change of Address — Please print new address below.Contents